University College - Chemistry Chem 112A Lecture Notes - Lecture 8: Nitrite, Without Loss Of Generality, Ph
20 views3 pages
13 Dec 2016
School
Department
Professor
Document Summary
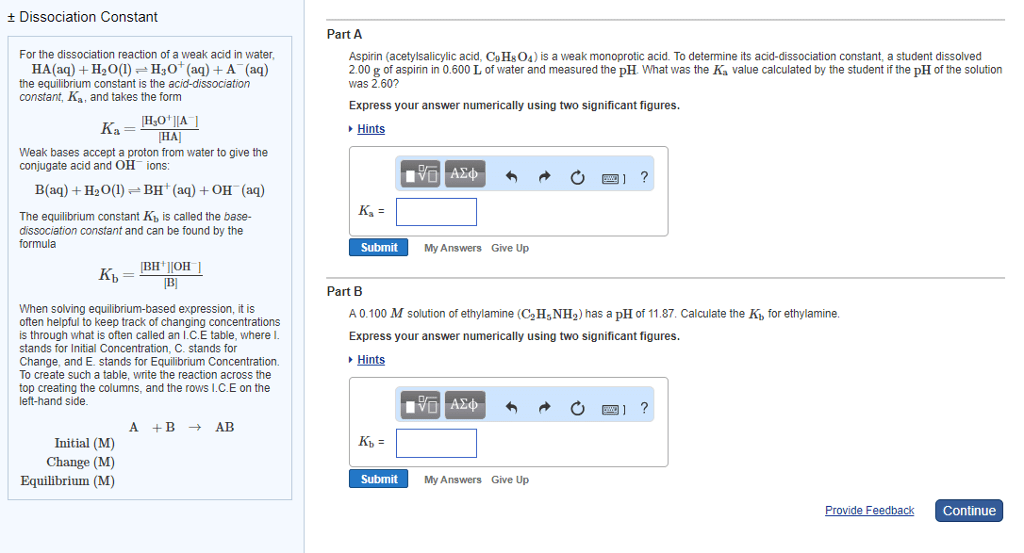
Equation of the day: =(cid:3028)(cid:3029: water and ph (aq) + a- (aq), with ka (aq) + ha(aq), with kb (aq) + oh- (aq, autoionization of water and ph. Weak base ionization: a: for a weak acid, water is a br nstead base. Generally an acidic solution has ph < 7, and a basic solution has ph > 7 (from clicker) for an endothermic reaction, k increases with increasing temperature. Strong acid equilibrium: hi + h2o h3o+ + i-, ka is infinitely large. Conjugate base equilibrium: i- + h2o oh- + hi, kb is infinitely small: limiting case with the conjugate see-saw: strong acids and bases. I- in solution will be neutral and not basic. Kb = kw/ka = 2. 86 x 10 -11 (aq), ka = 3. 50 x 10-4 (aq) + hf(aq), kb = 2. 86 x 10-11. Acid/base and ph: calculating ph of weak acid in water, nitrous acid: hno2 + h2o h3o+ + no2.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232