CHEM 100 Lecture Notes - Lecture 16: Sodium Chloride, Molar Concentration, Intermolecular Force
Document Summary
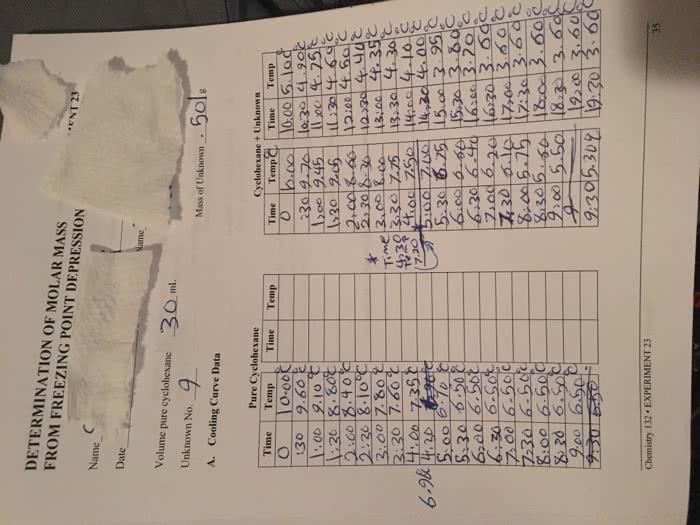
Identify a solute/solvent; types: factors that affect solubility, degree of saturation b. Intermolecular forces s/l like dissolves like i. g molar mass: pressure, temperature, qualitative analysis, molarity, molality, % concentration, dilutions, colligative properties, vp depression (vapor point, freezing point depression, bp increases, os. pressure increases. Saturated solution: solution containing the maximum possible amount of dissolved solute at the equilibrium. Henry"s law solubility= kp (constant depends on gas) Can either dissolve or crystalize the solution. Unsaturated + solute dissolved until equilibrium. As temperature increases the solubility or a solid in the solvent also increases. Most gases become less soluble in water as temperature rises. Molarity (m)= moles of solute/ liters of solution. Calculate molarity 4. 75 mol nacl to produce 3. 00 l solution. 4. 75 mol nacl/ 3. 00 l = 1. 58 m. Molality (m)= moles of solute/mass of solvent (kg) Percent by volume = volume of solute/ volume of solution (x) 100%