Use Table 10.1 in the textbook and figure 10.21 to answer the following three part question.
a) Calculate the ÎEºâ for the citrate cycle reaction converting malate to oxaloacetate and describe how you calculated this value.
b) What is the overall change in standard free energy (ÎGº') for this reaction and describe how you calculated it.
c) Do you think the actual change in free energy (ÎG) for this reaction differs significantly from the standard free energy (ÎGº') for the reaction, and if so, why is it different?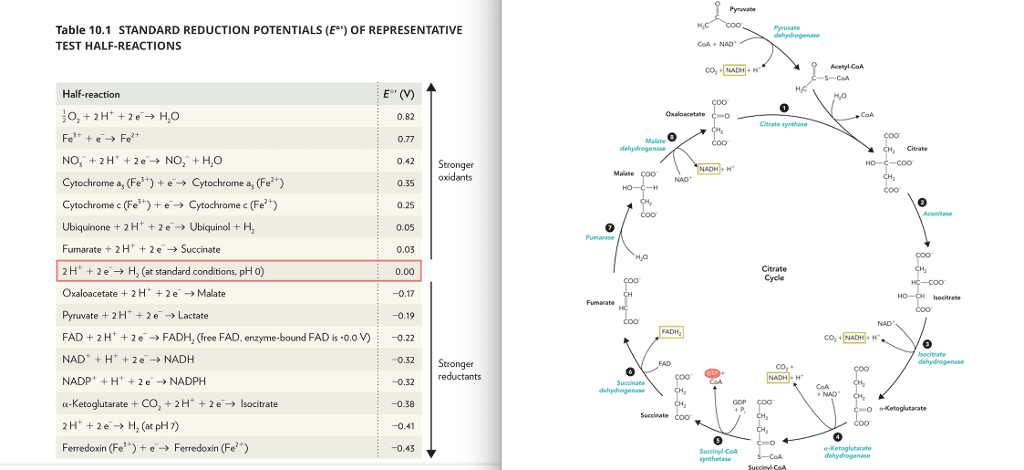
Table 10.1 STANDARD REDUCTION POTENTIALS (E) OF REPRESENTATIVE TEST HALF-REACTIONS CoA + NAD Half-reaction jo2 + 2H+ + 2e-âH2O r" + e-â Fe2+ NO, + 2H+ + 2 eâ No," + H2O Cytochrome as (Fe3+1+ e-â Cytochrome as (Fe2+) Cytochrome c (Felt) + e--> Cytochrome c (Fe2+) Ubiquinone + 2H+ + 2eâ Ubiquinol + H2 Fumarate 2H' + 2e--> Succinate 2H+ + 2 e â H, (at standard conditions, pH 0) Oxaloacetate + 2 H+ + 2 e--> Malate Pyruvate + 2H·+ 2e--> Lactate FAD +2H +2 FADH, (free FAD, enzyme-bound FAD is o.oV NAD" + H+ + 2e-â NADH NADP" + H + + 2 e'-> NADPH α-Ketoglutarate + CO2 + 2H' + 2e-â lsocitrate 2H' +2eâ H2 (at pH 7) Ferredoxin (Fe") + eâ Ferredoxin (Fe2+) Eo (V) 0 0.82 0.77 0.42 Stronger 0.35 0.25 0.05 0.03 0.00 Citrate sythase Cyele -0.19 022 0.32 Stronger -0.32 reductants 0.38 NAD 0.41 0,43




