CHEM 2320 Lecture Notes - Lecture 4: Ketone, Leaving Group, Mass Spectrum
Document Summary
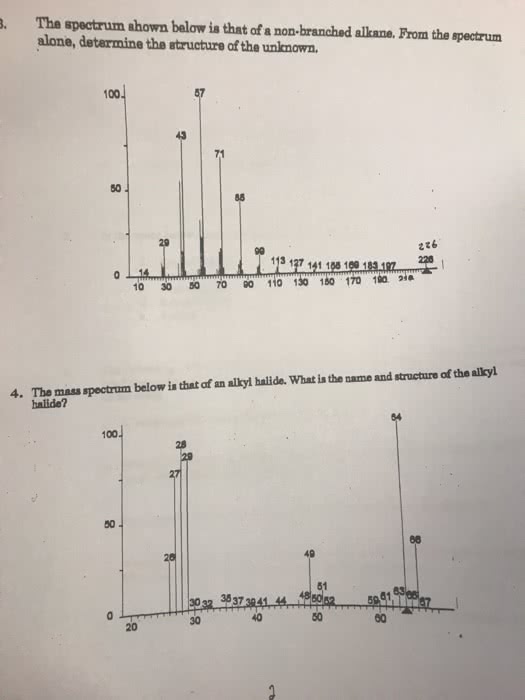
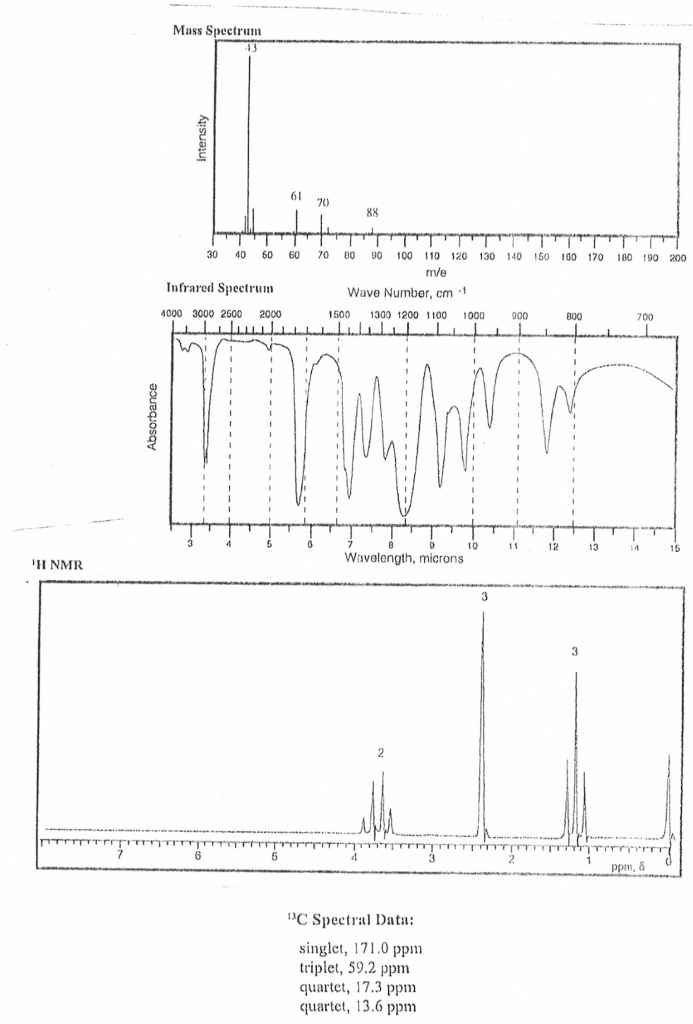
With is not branches a way that it as ie leads. Carbo cations regular break will in to the most. There are main peaks that we can identity but we can"t identity all of. Lower masses are not ot concern because they can be generated at almost any. +00 t isn"t it big so cation there in. When we see two peaks that are around. The same height typically it means that there"s. @ t. im#enesmmmpedabe for of carbon every number a@iy3} Mh for carbon we can estimate the carbons in a molecule. 1001 . can eutheaitterenu isotope patterns intensity of relative. I number of carbons is the rule of. 14+1 like does not need other ill in this have a. If it"s a big on any ions . have to be doesn"t to be. Bromine impact isolated molecular chlorine necessarily significant contamination fragment fragment calculations. M +2 the same a big there are.