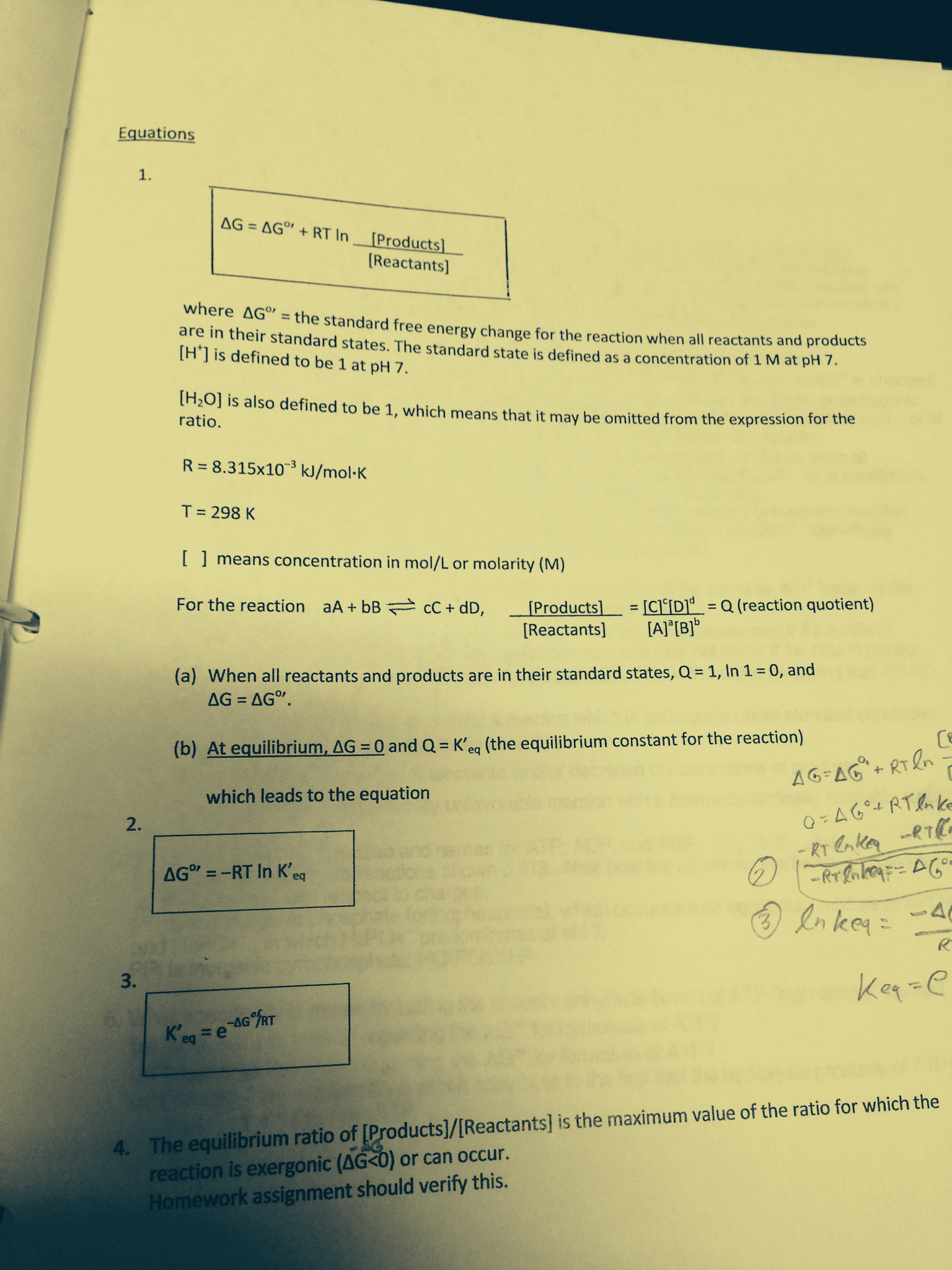CHEM 201 Lecture Notes - Lecture 27: Exergonic Reaction, Equilibrium Constant
11 views1 pages
Document Summary
While a reaction is actually occurring, the amounts of reactants and products are steadily changing and therefore q itself is changing, g is heading towards zero(equilibrium) and at zero, there is no further net change. G =-rtlnk: *k is the equilibrium quotient or the equilibrium constant, k is a specific condition of q at equilibrium q will equal k at equilibrium, at equilibrium, q=k, whereas under standard conditions, q=1. K=e- g/rt ln(k)=- g rt: the driving force represents nature"s drive toward maximum suniv, for any exergonic reaction, the greatest driving force (its most negative g) occurs at the moment of starting the reaction.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232