CHEM 2331H Lecture Notes - Lecture 9: Lone Pair, Ionic Bonding, Partial Charge
70 views2 pages
Document Summary
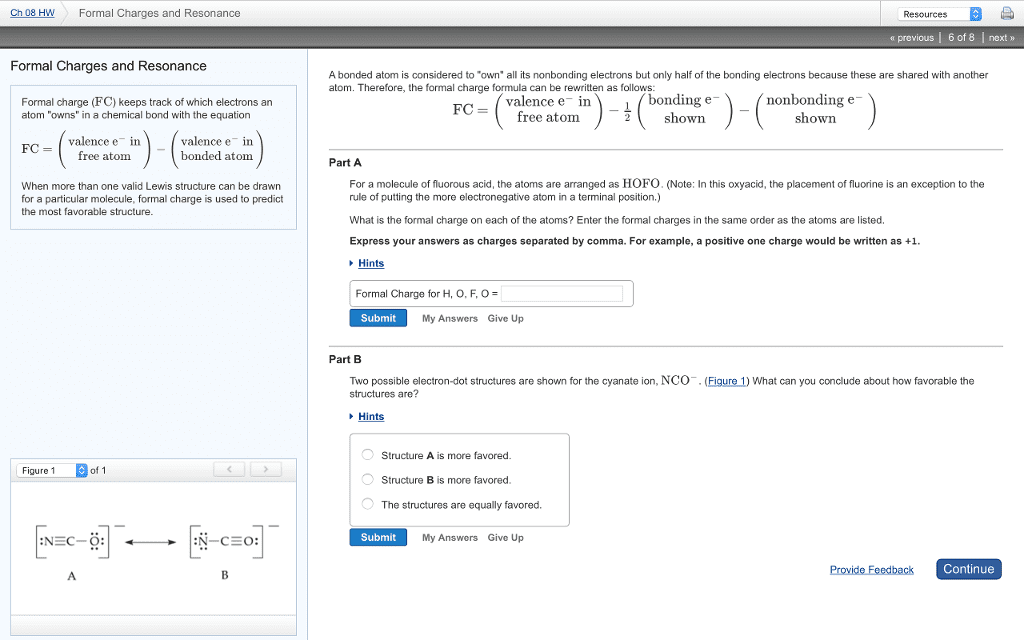
When electronegativity difference is too big, the more electronegative atom just takes the electron not sharing, transferring ionic bond. N normally owns 5 but in nh4, owns 4 5 4 = +1: ex: hydroxide: o has 1 bond and 3 lone pairs formal charge of -1. 6 7 = -1: tip: when # of bonds varies from typical valency, atom is probably charged, example: 1st one: carbon doesn"t have filled octet bad. 2nd one: oxygen is positive even though it is more electronegative . better to have carbon be positive. Consensus structure: graphical average of both structures. Use partial charges rather than exact fractions because haven"t weighted which resonance structures is better . don"t know which one is more dominant. Bond is also between length of single and double bond 1 solid bond and 1 dashed bond. Don"t draw lone pairs in consensus structures. Major and minor resonance contributors: resonance rules of thumb (with 1st being most important rule):
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232