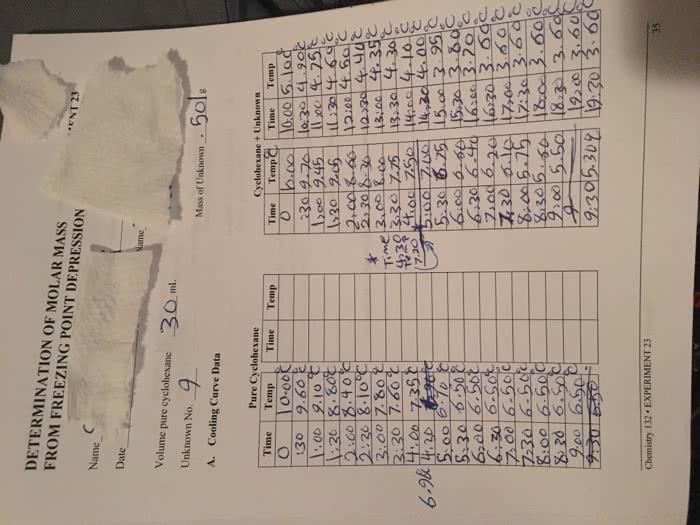
CHEM 1062 Lecture Notes - Lecture 4: Osmosis, Semipermeable Membrane, Boiling-Point Elevation
Document Summary
Osmotic pressure (vapor pressure lowering and boiling point elevation in lecture #3 notes and copied here) General note: these don"t dissociate and have negligible vapor pressure. Vapor pressure is always lower than the vapor pressure of the pure solvent ( p) Maintaining equilibrium: fewer solvent molecules on surface than solute, therefore fewer vaporize per unit time. To maintain equilibrium fewer molecules must enter liquid, which requires a lower vapor pressure. Increasing entropy: with dissolved particles present, the solution has a higher entropy so the solvent has a lower tendency to vaporize. Quantifying vapor pressure lowering: raoult"s law solvent solvent is the vapor pressure of the pure solvent. Psolvent is the vapor pressure of the solvent in solution. Xsolvent is the mole fraction of the solvent. An ideal solution is a solution which follows raoult"s law exactly. A solution boils at a higher temperature than a pure solvent. This is a result of the vapor pressure lowering.