CHEM 1062 Lecture Notes - Lecture 20: Sodium Acetate, Acetic Acid, Conjugate Acid
82 views2 pages
Document Summary
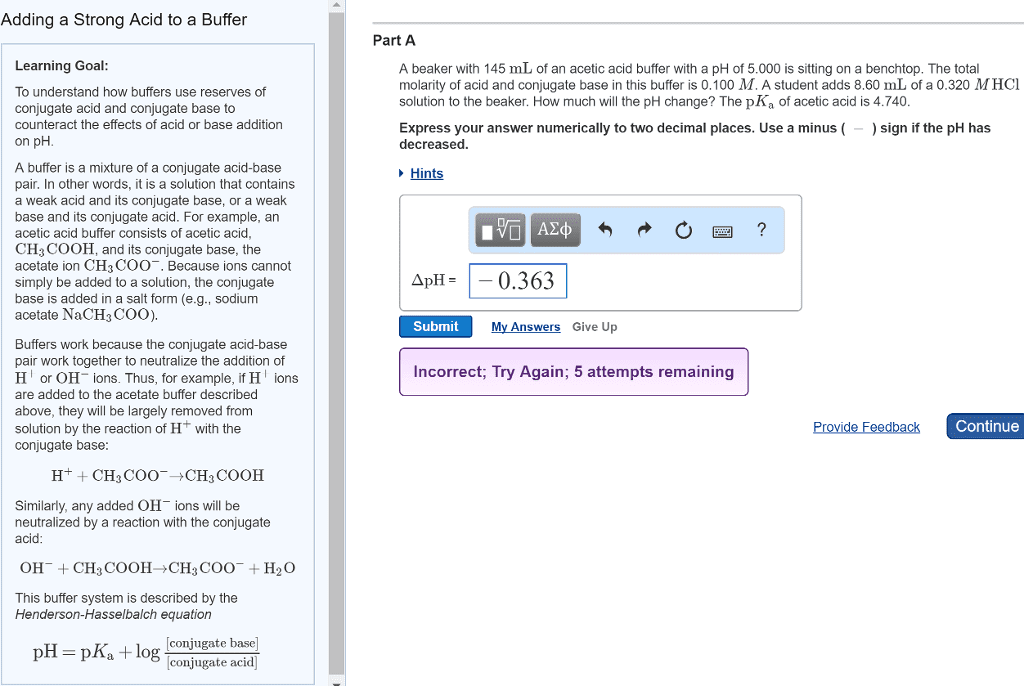
Buffer: a solution that contains a mixture of a weak acid and its conjugate base and resists changes in ph by neutralizing. Ch3coo- + h3o+ ch3cooh + h2o. Add h3o+ to ch3coo-, neutralizes the h3o+ . Contains mainly ch3cooh (very weak so it doesn"t dissociate much) Ch3cooh + oh- ch3coo- + h2o. Add oh- to ch3cooh, neutralizes the oh- . Mixture of ch3cooh and ch3coo- (this is a buffer) Contains mostly ions of ch3cooh and ch3coo- Add h3o+, ch3coo- neutralizes it, producing extra ch3cooh. Add oh-, ch3cooh neutralizes it, producing extra ch3coo- Important: as long as the buffer contains enough ch3cooh and ch3coo-, it neutralizes added. H3o+ or oh- without appreciable change in composition (especially ph) Q: find ph of a buffer that has 0. 5 m ch3cooh (ka = 1. 8*10-5) and 0. 50 m ch3coona. 0. 50 +x ~ 0. 50 ~ 0. 50 - x therefore: 1. 8*10-5 = x = [h3o+] ph = -log[h3o+], ph = -log (1. 8*10-5) ph = 4. 74.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
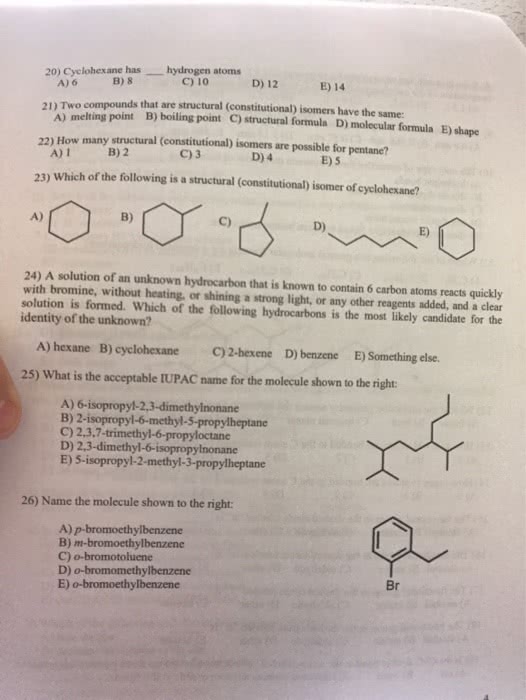
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232