CHEM 1062 Lecture Notes - Lecture 1: Miscibility, Sodium Chloride, Freezing-Point Depression
28 views3 pages
Document Summary
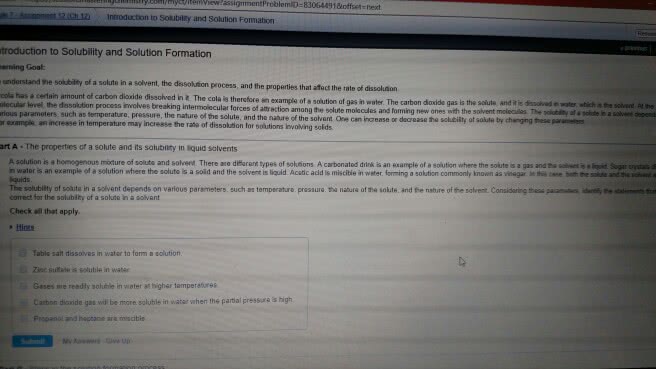
Solution = homogeneous mixture of 2 or more chemicals, 1 phase uniform distributions of molecules throughout. Gas in a liquid = oxygen in water. Solid in liquid = salt in water (ionic) Gas in gas = air; all gases infinitely soluble in each other. Colloidal suspensions = not solutions, very stable suspensions of very fine particles (nanometer) Do not settle in the liquid because of brownian motion of liquid molecules. Example: milk, nano particles of fat & proteins. Molarity (m) = number of moles of solute / volume usually in l of solution (total volume) Molality (m) - moles of solute / mass of solvent ; this does not depend on temperature. Percent- weight by weight = (mass of solute / mass of solution) * 100. Mass of solution = mass of solute + mass of solvent. Percent- volume by volume = (volume of solute / volume of solution) * 100.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232