CHEM 111 Lecture Notes - Lecture 4: Dmitri Mendeleev, Relative Atomic Mass, Mass Number
Document Summary
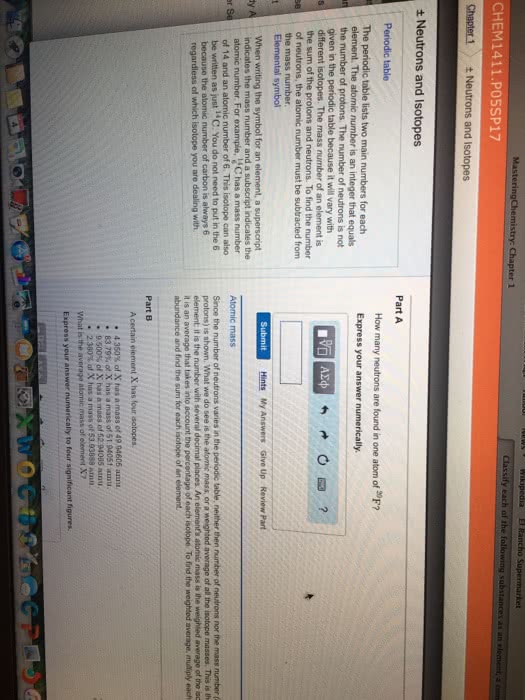
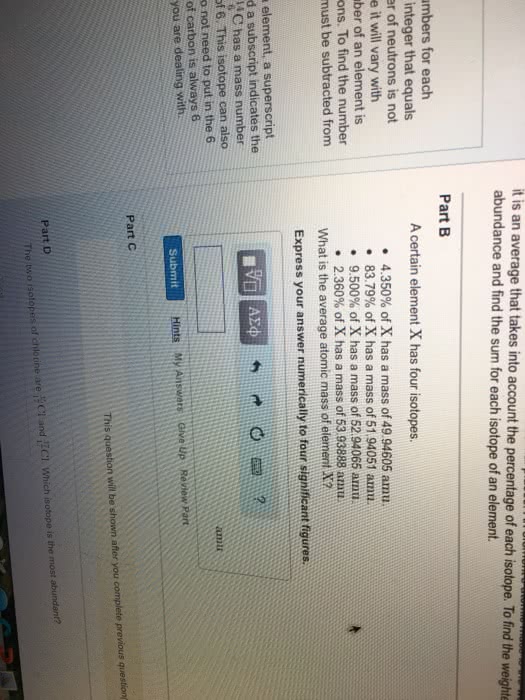
We use abbreviations called atomic symbols to describe elements (ex. carbon= c) Atomic number is equal to the number of protons in the element. All atoms of the same element have the same number of protons. In neutral atoms, the number of protons= number of electrons. It is equal to the number of protons + number of neutrons. Mass number can change for different atoms of the element (ex. Isotopes: atoms with the same atomic number (same number of protons) but different mass number (different number of neutrons) The the total percentage abundance of all isotopes of an element should equal 100% Atomic weight: due to the existence of isotopes, the mass of a collection of atoms of an element has an average value. Atomic weight = (% abundance of isotope a/100) x mass of isotope a + (% abundance of isotope b/100) x mass of isotope b +etc.