CHEM 6A Lecture 14: CHEM W5 Lec 11/1/17
47 views3 pages
3 Jun 2018
School
Department
Course
Professor

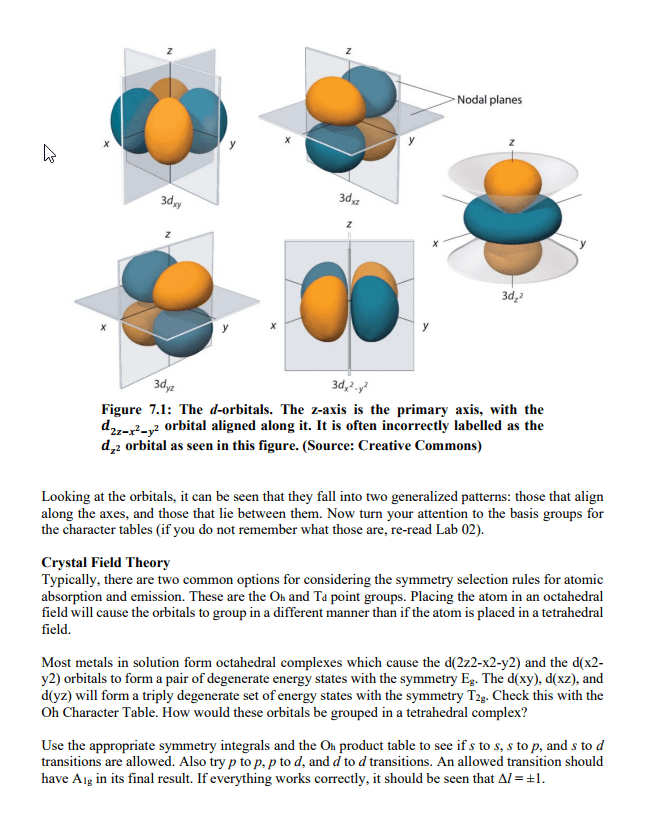
Stationary States in Bohr’s Model of H Atom
● Note that the energy differences between stationary states are NOT the same!
● The stationary states become closer together as n increases
Emission Lines According to Bohr’s Model
From nucleus of a
hydrogen atom
Allowed Energy Levels for Electrons
● H atom: En = -2.1799 aJ , n = 1, 2, 3...
n2
● Any ion with one electron and Z (atomic number) > 1
○ En = -2.1799 (Z2/n2) aJ, n = 1, 2, 3…
● E = 0, when n =∞
○ Therefore En below n =∞(e- is ionized) have negative values
● Lower energy = more stable
● Ground state = most stable state
Unlock document
This preview shows page 1 of the document.
Unlock all 3 pages and 3 million more documents.
Already have an account? Log in


19
CHEM 6A Full Course Notes
Verified Note
19 documents
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232