CHEM 1127Q Lecture Notes - Lecture 9: Chemical Equation, Solution, Chlorate
24 views3 pages
1 Mar 2019
School
Department
Course
Professor

44
CHEM 1127Q Full Course Notes
Verified Note
44 documents
Document Summary
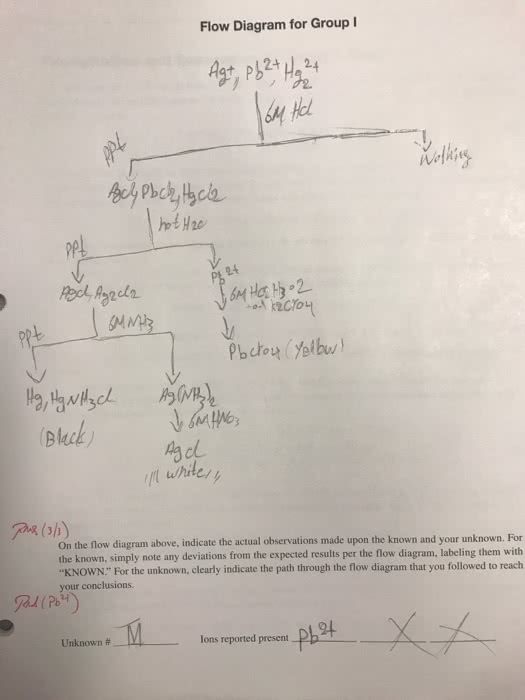
A precipitation reaction is one in which dissolved substances react to form one or more solid products. A compound is soluble in a particular liquid if it dissolves in that liquid. A compound is insoluble if it does not dissolve in the liquid. An aqueous solution is a homogenous mixture of a substance with water. When ionic compounds dissolve in water, they usually dissociate into their component ions. Group 1 metal cations and ammonium ion. Halides of ag+, hg2 2+, and pb 2+ Sulfates of ag+, ba2+, ca2+, hg2 2+, pb 2+, sr2+ Compounds of these anions with group 1 metal cations and ammonium anion. Hydroxides of group 1 metal cations and ba2+ A molecular equation is an equation showing the complete neutral formulas for every compound in the reaction. Complete ionic equations show aqueous ionic compounds that normally dissociate in solution as they are actually present in solution.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232