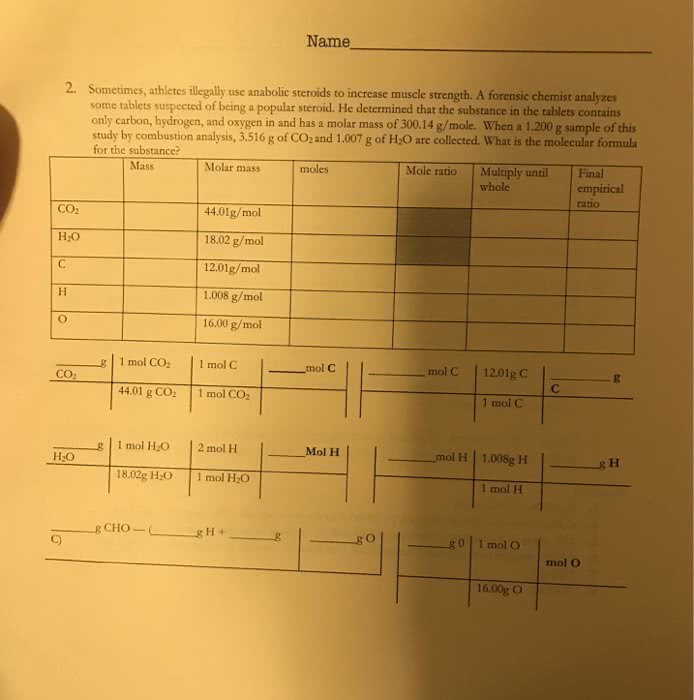
CHEM 1122 Lecture Notes - Lecture 23: Chemical Formula, Empirical Formula, Molecular Mass
Document Summary
Empirical formula is the simplest whole number ratio of atoms in the chemical formula. Molecular formula is the real number of atoms in the molecule. 0. 8563 g c x 1 mol c/12. 011 g c = 0. 07129 mol c. 0. 1437 g c x 1 mol h/1. 0078 g h = 0. 1426 mol h. Divide both subscripts by lowest value (0. 07129) = ch 2: start with assumption of 1000 g. Mass % value will translate directly to gram amount: calculate moles for each atom. For mass %, oxygen amount is calculated by difference: divide by the smallest number of moles. 0. 2668 g c x 1 mol c/12. 011 g c = 0. 02221 mol c. 0. 0224 g h x 1 mol h/1. 0078 h = 0. 02221 mol h. 0. 7108 g o x 1 mol/15. 999 g o = 0. 0443 mol o. After dividing by smallest number of moles, we get: cho 2. 0. 2592 g x 1 mol n/14. 067 g h = 0. 018514 mol n.