CHEM 17 Lecture Notes - Lecture 6: Chemical Formula, Formula Unit, Molecular Mass
Document Summary
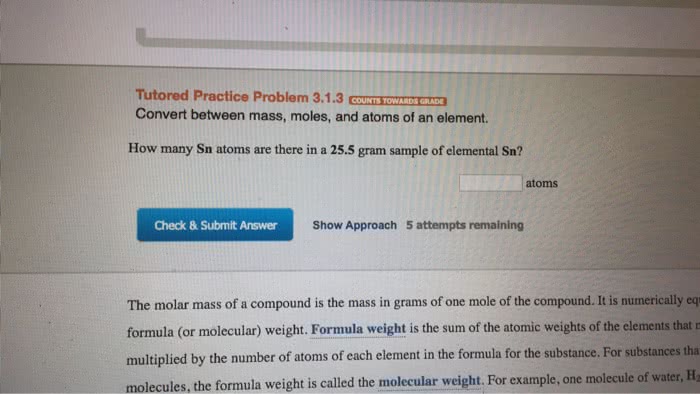
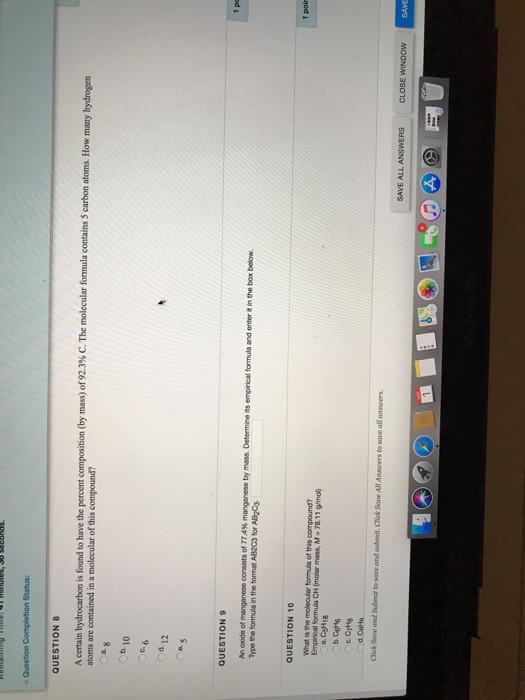
Lecture 6 and 7- balancing equations, mole calculations. The sum of the atomic weights of all the atoms in a molecule of the substance. The sum of the atomic weights of all atoms in a formula unit of the compound whether molecular or not. The quantity of a given amount of substance that contains as many molecules or formula units as the number of toms in exactly 12 g of carbon 12. The number of atoms in exactly 12g of carbon 12. Carbon 12 has a molar mass of 12g/mol. The mass percentage of each element in the compound. It helps determine the formula of new compounds. The mass percentage of a in a substance is the parts of a per hundred parts of the total by mass. Mass % a = mass of a in the whole/mass of the whole * 100% The formula of a substance written with the smallest integer subscripts.