CHEM 14CL Lecture Notes - Lecture 5: Sodium Sulfate, Miscibility, Ethyl Acetate
Document Summary
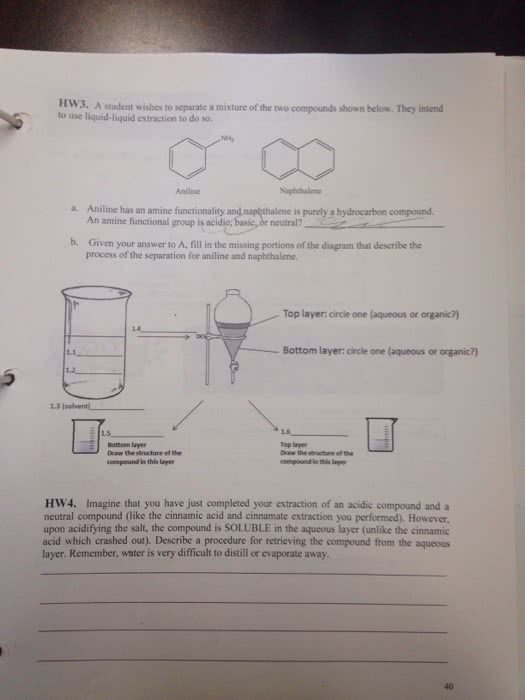
Isolate a substance in the first solvent second solvent which is immiscible with the shake the solution with a first separators tunnel. Cap ( thatmust be removed when draining timid a. Mose to drain solvent also need to open. D off glass will shatter organ to later is non. Polar aqueous layer denser layer is is assumed for this course polar drained usually organic layer is on top ) underlying principle in extraction distribution law : a substance ( s ) distributes itself between. 2 immiscible solvents so that the ratio of the concentration in one solvent to the concentration in the second solvent always remain the same c @ constant temp a. C aka kp ) extraction equation finding amount of materials remaining insolvent. Wi ( any v. n n =l n > 1 for for single extraction. X= v vz initial amount of substance being extracted partition coefficient volume volume of of solvent.