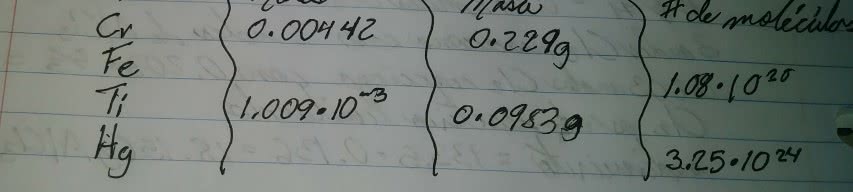CHEM 001A Lecture Notes - Lecture 6: Barium Chloride, Solvation, Stoichiometry
47 views2 pages
8 Mar 2018
School
Department
Course
Professor
Document Summary
Chem 1a 10/19/17: solution concentrations, molarity (m) = mol/l, # mol = v*m , solution preparation, solution stoichiometry, the role of water as a solvent. Solutions: solutions are defined as homogeneous mixtures of two or more pure substances, the solvent is present in greatest abundance, all other substances are solutes, when water is the solvent, the solution is called aqueous solution. Molarity: molarity (m) = moles (n) of solute / volume (v) of solution in liters, m = n / v. If we know the volume (v) and the molarity (m) of a solution, we can calculate moles (n: n =v x m. Solvation: remember the rule: likes dissolve likes. 5 l x 0. 0525 m = . 02625 mol: al(no3)3 = 27. 0 g al + 3 (14. 0 g n) + 9 (16. 0 g o) = 213. 0 g/mol, 0. 2625 mol x 213. 0 g/mol = 5. 59 g.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232