CHEM 001A Lecture Notes - Lecture 8: Molar Mass, Ionic Compound, Formula Unit
Document Summary
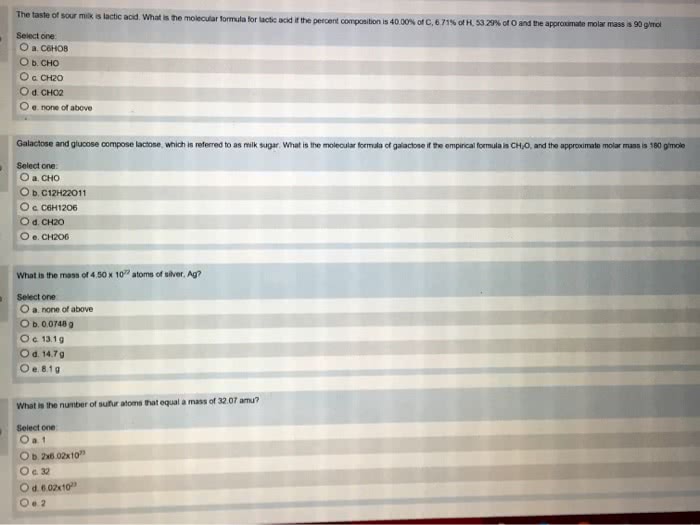
One mole- defined as the number of atoms in exactly 12 grams of carbon-12: used as a easy way of grouping a number of particles, 6. 022e23 particles/mole. Molar mass: used to find mass (in g/mol) equal to mass of element, or molecule (in amu/particle, one atom of he=4. 003amu. 1 mole of he equal to 6. 022e23 atoms. Molecular mass: sum of atomic masses of the elements in the compound, co2 = c + 2o, co2 = 12. 01 + 16(2, co2 = 44. 01 amu/molecule. Formula mass: mass in atomic mass units of one formula unit of an ionic compound, nacl = na + cl, nacl = 22. 99 + 35. 45, nacl = 58. 44 amu/molecule (amu/molecule is the same as g/mol) Percentage composition- percentage by mass of each element in a component: percentage composition of carbon in ch4, c/ch4= 12. 01/16. 05 x 100%= 74. 88% C1v1=c2v2, where c is concentration and v is volume.