37.
A/ What types of crystals are formed by the following solid elements and compounds: Ca, CaF2 , and CF4 ? In each case, state the type of forces among the. particles that make up the lattice.
B/ When ice melts, some individual water molecules become non-hydrogen-bonded. Using this information and Figure 7.4b, speculate on why the density of liquid water is greater than that of ice.
Figuer 7.4: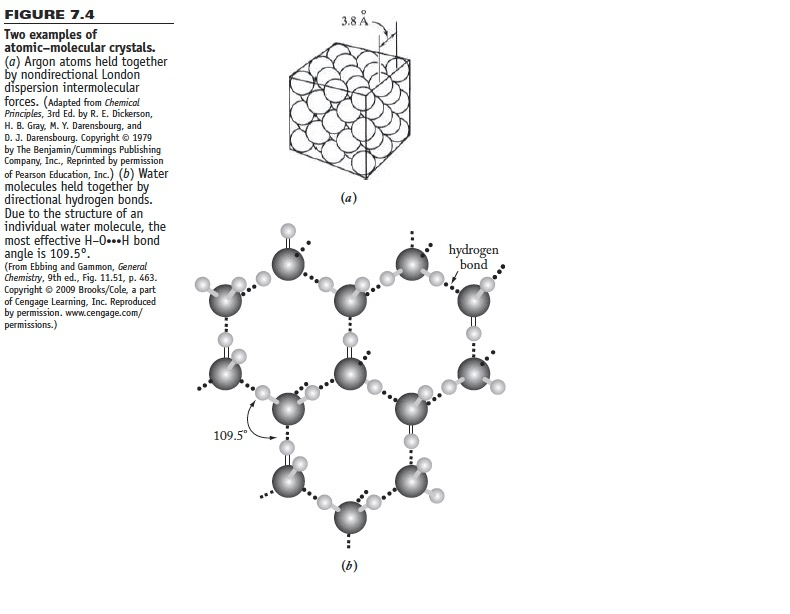
please explain we have an exam on monday as well; will rate real quick
FIGURE 7.4 Two examples of atomic-molecular crystals. a) Argon atoms held together nondirectional London dispersion intermolecular forces. (Adapted from chemical Principles, 3rd Ed. by R. E. Dickerson, H. B. Gray, M. Y. Darensbourg, and D. J. Darensbourg. Copyright 1979 by The Benjamin/Cummings Publishing Company, Inc., Reprinted by permission of Pearson Education, Inc.) (b) Water molecules held together directional hydrogen bond Due to the structure of an individual water molecule, the most effective H-0...H bond angle is 109.5. (From Ebbing and Gammon, General Chemistry, 9th ed., Fig. 11.51, p. 463. Copyright 2009 Brooks/Cole, a part of Cengage Learning, Inc. Reproduced by permission. www.cengage.com/ permissions. 109.50 3,8 A (a) (b) hydrogen bond



