CHEM H90 Lecture Notes - Lecture 13: Steric Number, Vsepr Theory, Molecular Geometry

More about Lewis Structures
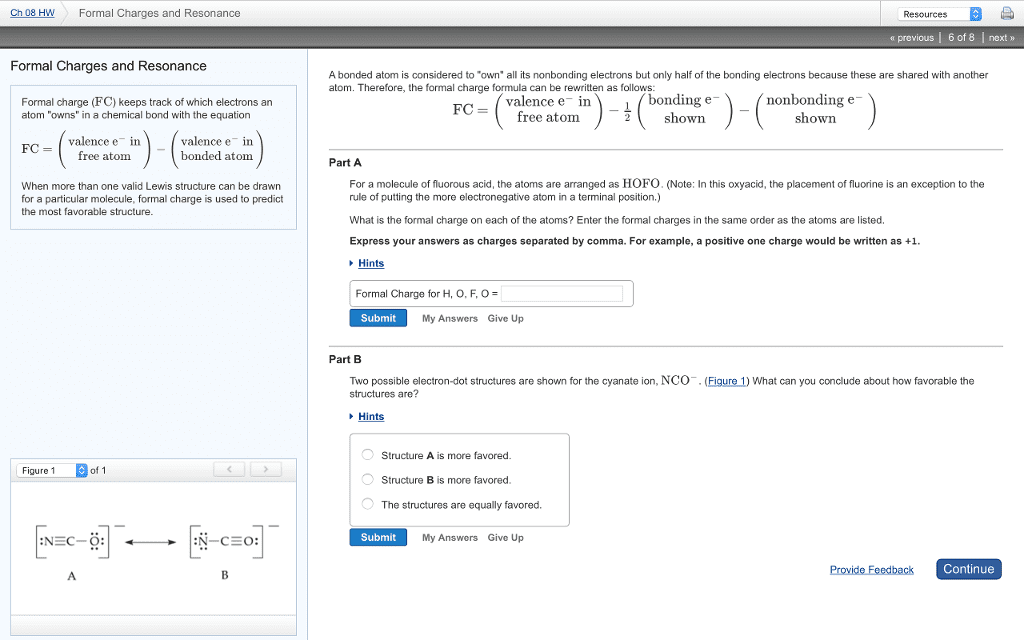
• resonance
o 2 or more structures that are both equally probable
o some molecules cannot be represented by one Lewis structure alone. two or more
resonance structures are required to describe the electron distribution
• exceptions to octet rule
o octet rule applies mainly to second period elements. for other elements, many
exceptions exist due to
▪ incomplete octet: there are not enough valence electrons available in the
structure to fill the octet on each atom
▪ expanded octet: octet rule based on filling s and p shells (2+6=8
positions). heavy atoms have d shells available and thus more positions to
fill electrons
o incomplete octet
▪ some atoms lack the number of valence electrons to fill the shell. they
form stable compounds with less than eight electrons surrounding them
▪ typical elements: Be, B, Al
o expanded octet
▪ elements beyond period 2 may have more orbitals available for allocating
electrons. this often results in atoms surrounded by more than 8 electrons
▪ notable examples: P, S, As
• VSEPR theory
o valence shell electron pair repulsion (electron domain model)
o because electrons are negatively charged, they repel each other
o in a molecule, bonds and lone pairs stay as far apart as possible
o rules
▪ the arrangement of bonds and loen pairs around the central atom is
controlled by electrostatic repulsion
▪ arrange all bonds and lone pairs as far apart as possible
▪ double and triple bonds can be treated approsimately like single bonds.
count them all, that’s the steric number, and determine basic shape
▪ if a molecule has resonance structure, VSEPR can be applied to all of
them
▪ number of attachment to central atom = steric number
• distortions
o lone pairs (or double bonds) have more electron density, and repel other bonds a
little, so angles between bonds shrink
o call it distorted
o instead of AXSN, write ABCNELPN
o e.g. H20: 0 has SN=4 for two bonds and two LP’s, so CN=2
o call it distorted
• rules for predicting molecular geometry
o write the lewis structure
o assign an electron group arrangement
o predict the ideal bond angle from the electron group arrangement (SN)
o predict deviations caused by lone pairs or multiple bonds
find more resources at oneclass.com
find more resources at oneclass.com