CHEM 51C Lecture Notes - Lecture 5: Infrared Spectroscopy, Chemical Shift, Chemical Formula
48 views5 pages
Document Summary
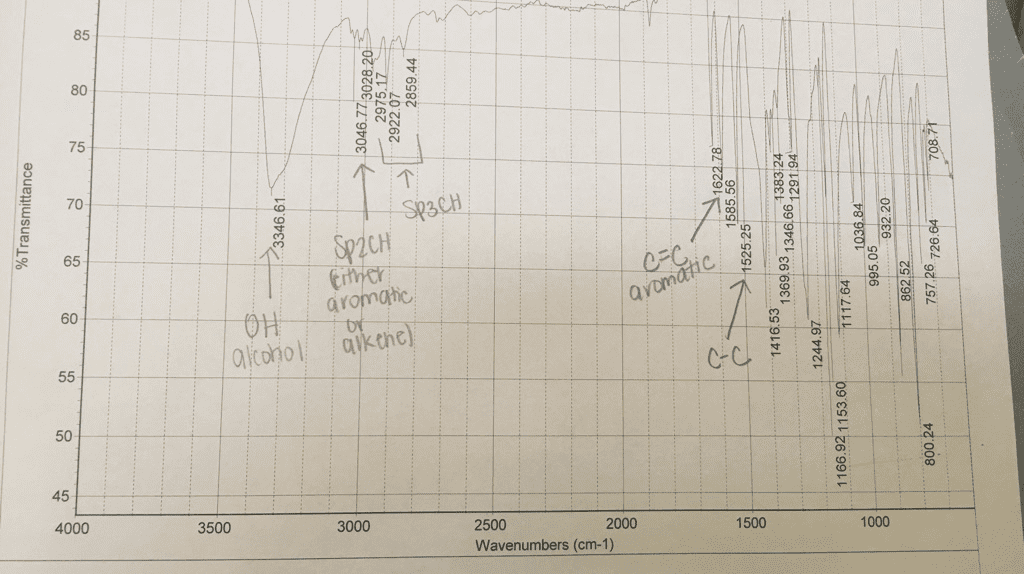
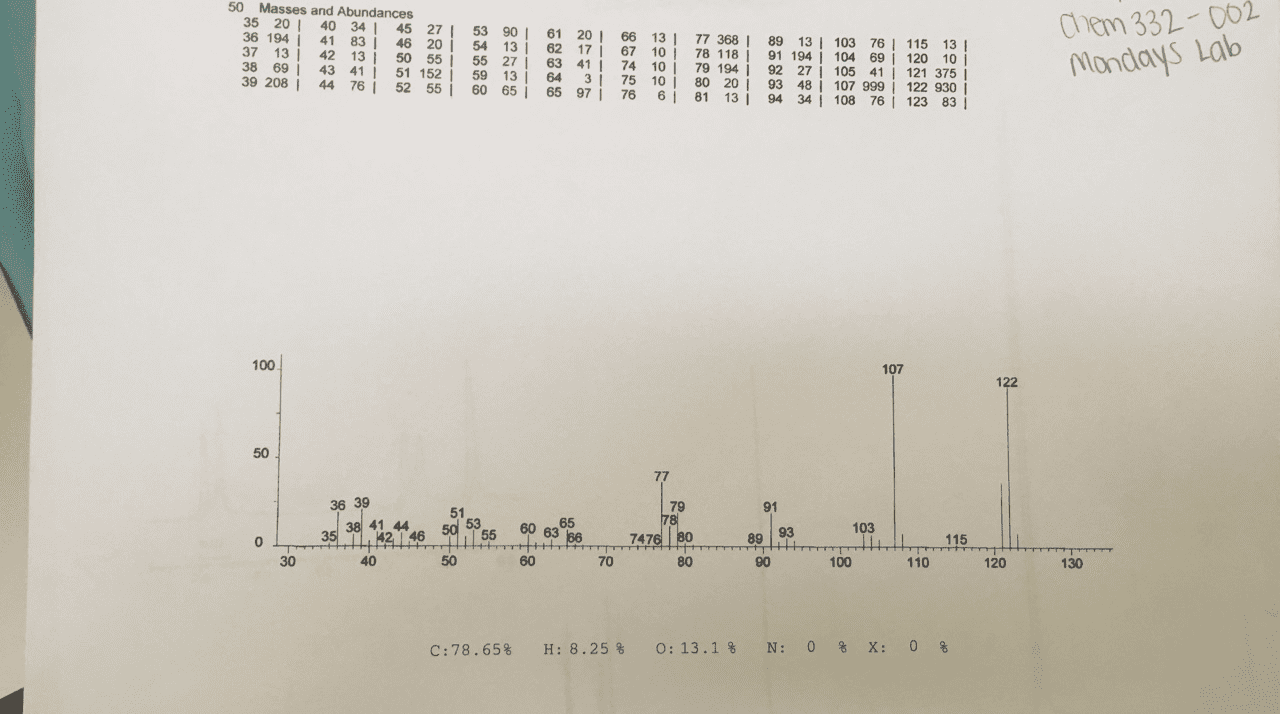
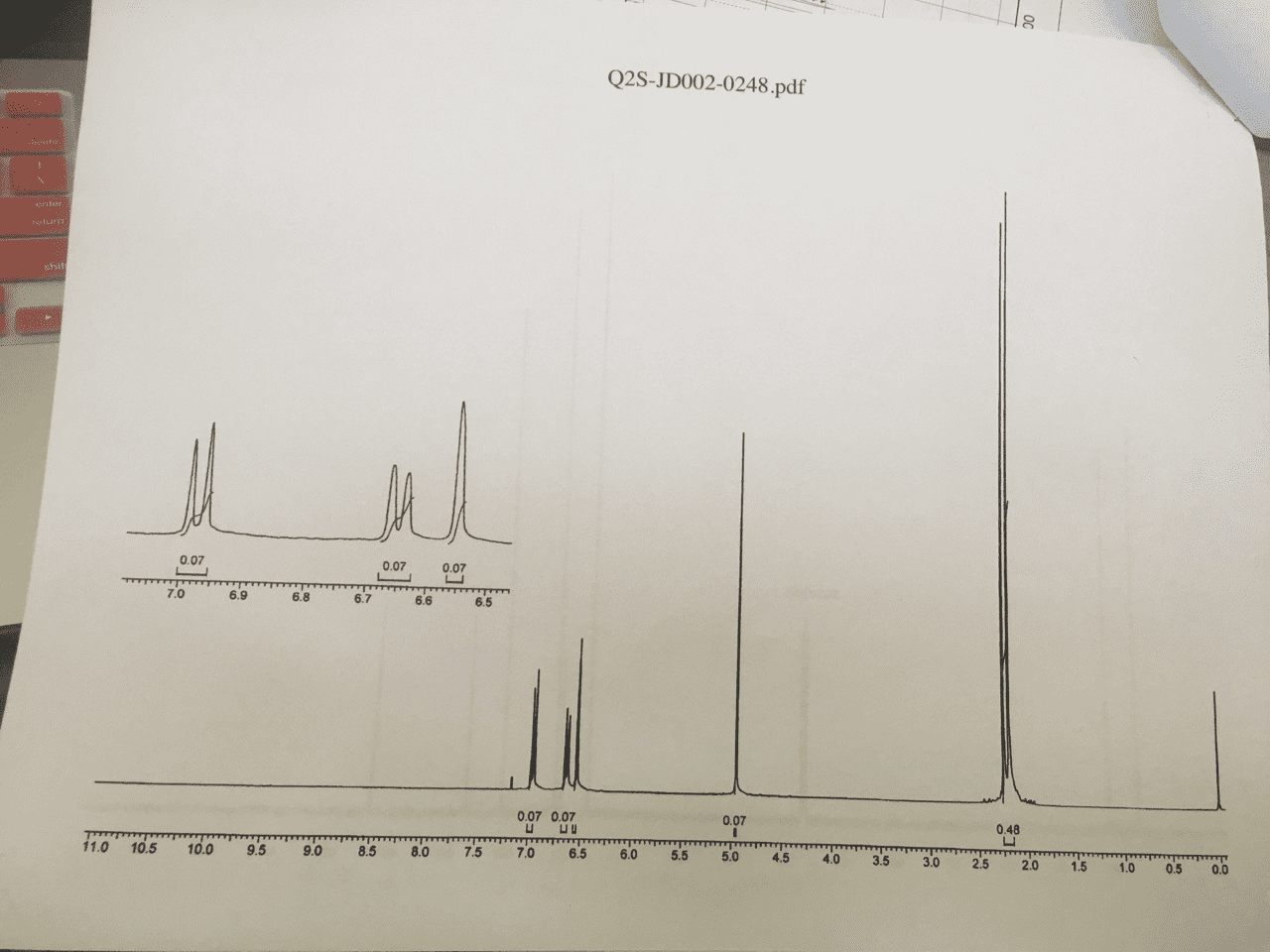
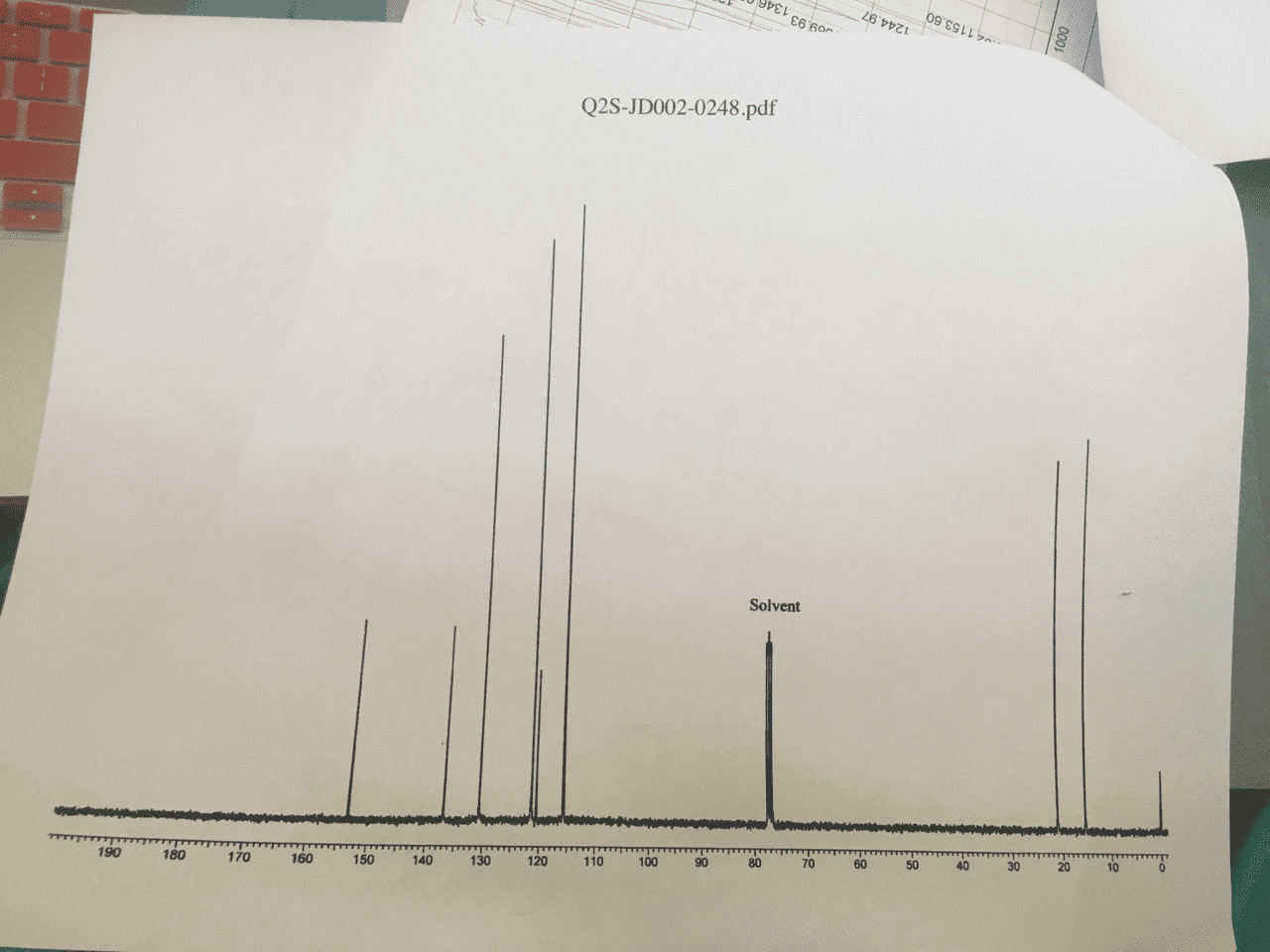
Look at molecular formula: count the carbons on the cnmr, symmetrical carbons would have the same signal. Look at hnmr for integrations, chemical shift and splitting. Compare the molecular formula to the alkane: cnhn+2: count number of carbons in molecular formula, count number of hydrogens a. If you have an o atom in the molecular formula, ignore it b. If you have a halide [f-, br-, cl-, i-], 1 halide= 1 h c. 1 ring = 1 degree of unsaturation a. ii. 1 double bond= 1 pi bond = 1 degree a. iii. 1 triple bond = 2 pi bonds= 2 degrees of unsaturation. 110-0= sp3 hybridized carbons [carbon bonding to 4 substituents] Key things to look for: multiplet in 6. 5-8 ppm- benzene ring, a doublet splitting (6 hs) , a singlet peak (9hs)= three methyl groups bonded to a carbon, singlet peak around 1-5: -oh group.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232