Fill the questions according to Data Table 7-2
5. balanced formula equation: for each combination of solutions that produced new product. write correct chemical formulas for the new compounds that could form from the ions present. Indicate which of the new compunds is a precipitate. If no reaction occurs, write NR.
6. complete ionic equation: write complete ionic equations for the precipitation reactions that you observed. show the reactants as ions, the precipitate as a solid, and the spectator ions as unchanged. balance each equation for mass and change. the total charge should be zero on each side of the equation.
7. net ionic equation: write the net ionic equations for each reaction in the preceding question.
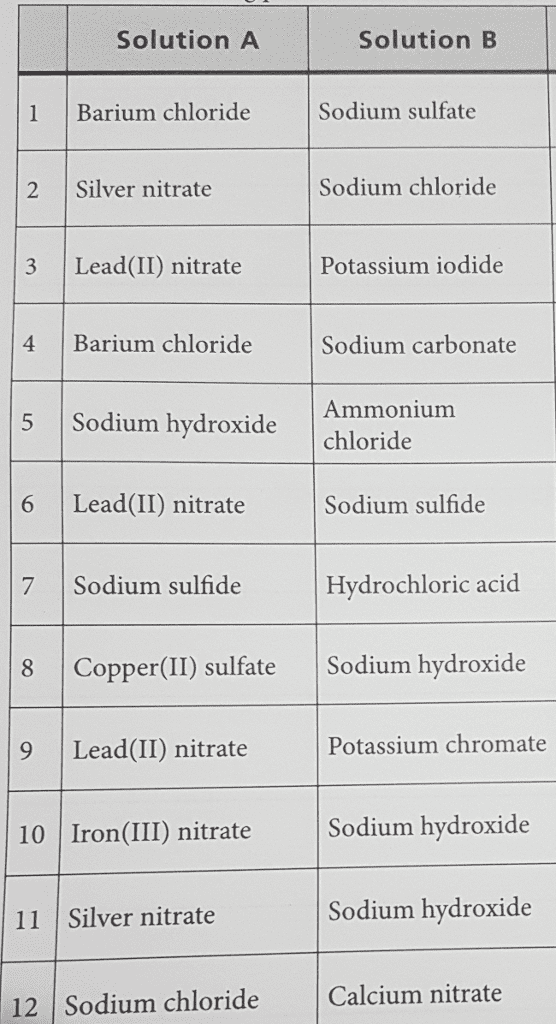


Solution A Solution B Sodium sulfate Barium chloride Sodium chloride 2 Silver nitrate 3 Lead(II) nitrate Potassium iodide 4 Barium chloride Sodium carbonate 5 Sodium hydroxide Ammonium chloride 6 Lead(II) nitrate Sodium sulfide Hydrochloric acid 7 Sodium sulfide 8 Copper(II) sulfate Sodium hydroxide Potassium chromate 9 Lead (II) nitrate Sodium hydroxide 10 ron (III) nitrate Sodium hydroxide 11 Silver nitrate 12 sodium chloride Calcium nitrate






