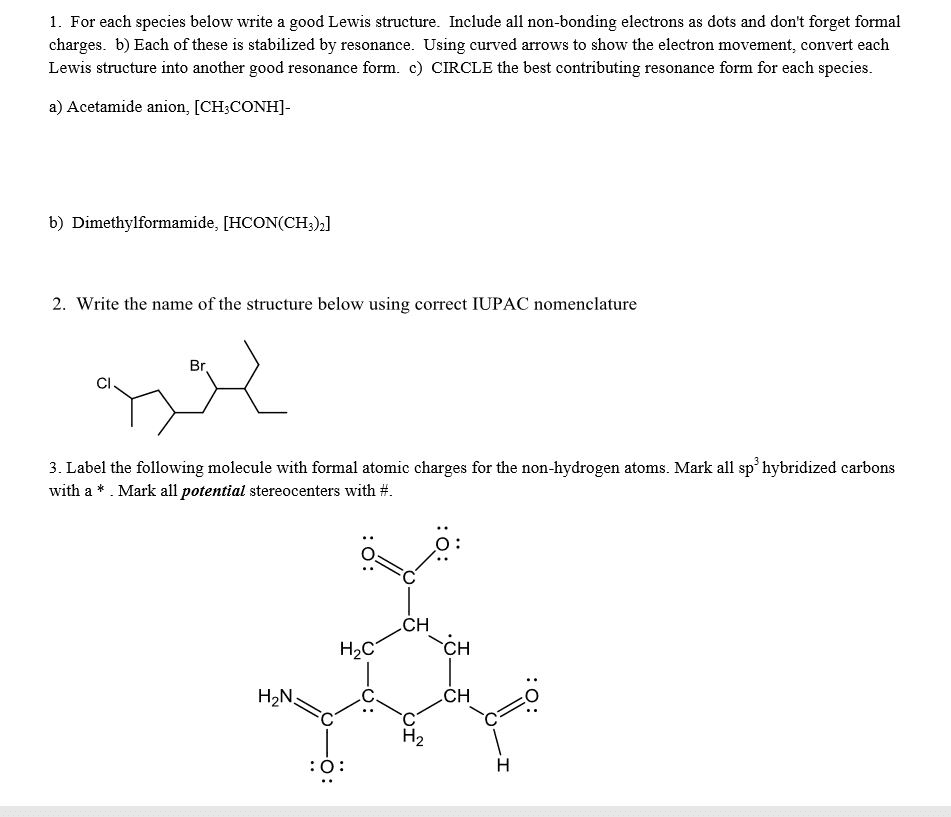
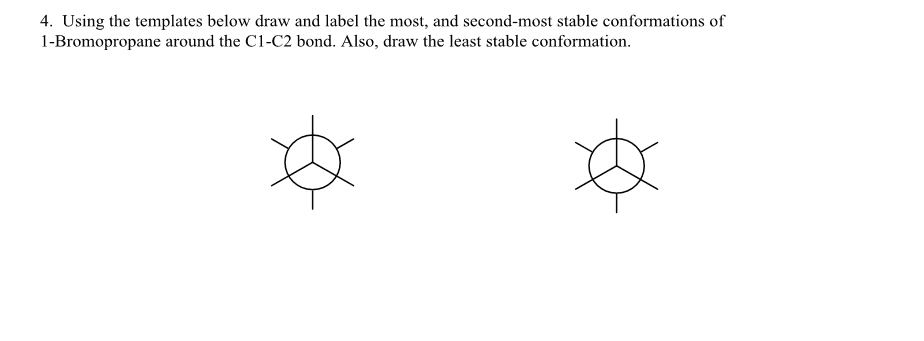
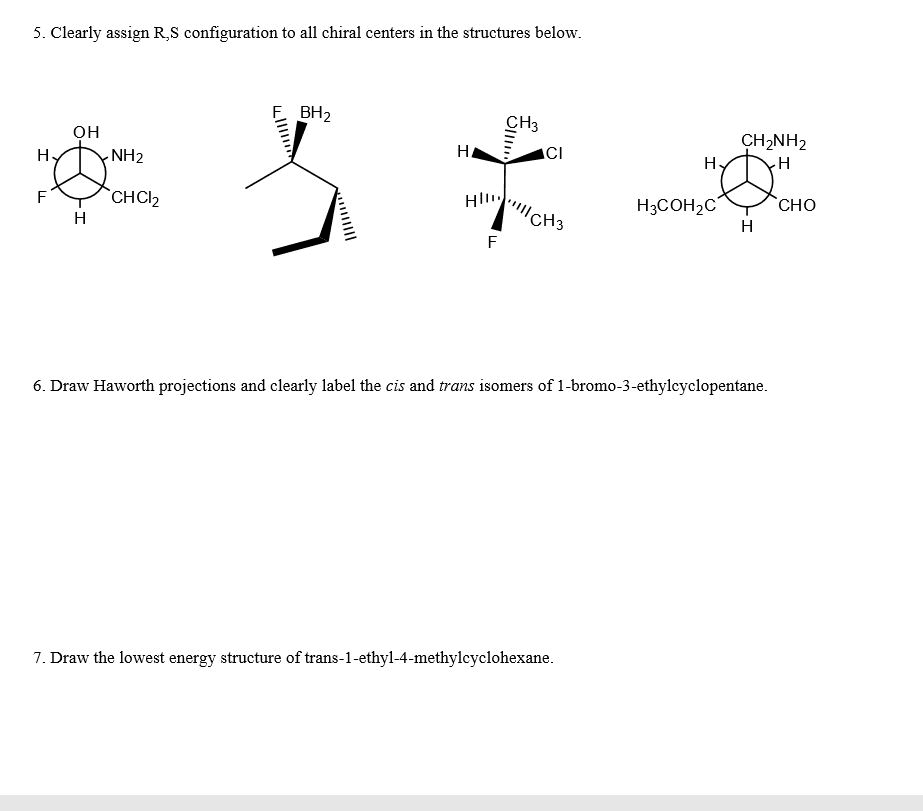
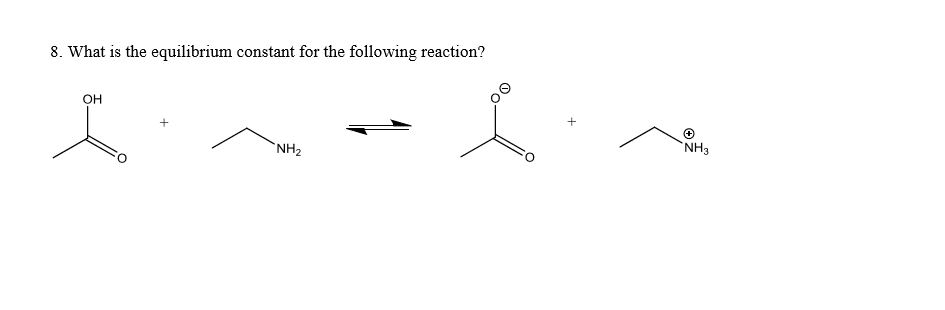
Spreading of positive or negative charge over two or more atoms in a compound is called __. isomerism delocalization stereoisomerism localization none of these Delocalization of charge over two or more atoms __ a molecule. destabilizes de localizes localizes stabilizes none of these Resonance structures have connectivity of atoms and __ distribution of electrons. different, same same, same different, different same, different none of these Which of the following violates the rules for curved arrows? I II IV I & III Ill & IV None of these Which of the following violates the rules for curved arrows? I & Il III & IV l. Il & III II, Ill & IV All of these Which of the following is a correct resonance structure for compound A? I Il III IV None of these Which of the following is/are the most significant resonance structure(s)? I II Ill I & II l & IV A Bronzed-Lowry acid is defined as a and a Bronzed-Lowry base is defined as a __. proton acceptor, proton donor proton donor, proton donor proton acceptor, proton acceptor proton donor, proton acceptor none of these for the following reaction, which reactant functions as an acid? I II neither which of the following is not a Bronzed-Lowry acid? CH_3OH CH_3CH_3 CH_3NH_2 CH_3SH None of these In a Bronzed-Lowry acid-base reaction the product(s) is (are) a __. Lewis acid conjugate acid only Lewis acid-base pair conjugate acid-conjugate base pair none of these A losses of proton from a Bronzed-Lowry acid results in a __. Lewis acid conjugate acid conjugate base conjugate acid-base pair none of these the tail of the curved arrow must start at __ electron density and the head must end at electron density. low, high high, high low, low high, low which of the following compounds is most acidic? I II Ill IV None of these