CHM 1142 Lecture 6: Chapter 6 - Electronic Structure of Atoms
Document Summary
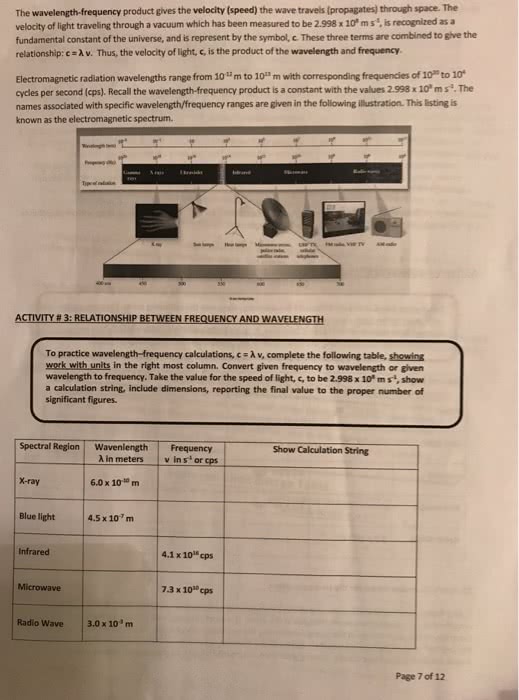
Electronic structure - arrangement and energy of electrons. To understand the electronic structure of atoms, one must understand nature of electromagnetic radiation. Wavelength = distance between corresponding points on adjacent waves. Speed of light: (c) = 3. 00 x 10^8 m/s. Red 7 x 10^-7 = largest wavelength and short frequency. Purple 4 x 10^-7 = shortest wavelength and largest frequency. Frequency (nu) = speed of light (c) / wavelength (lambda) Wavelength (lambda) = speed of light (c) / frequency (nu) The wave nature of light does not explain how an object can glow when its temperature increases. Max planck explained it by assuming that energy comes in packets called quanta. Matter can emit and absorb energy only in whole number multiples. Einstein used quanta to explain: photoelectronic effect - emission of electrons from an atom or molecule by absorption of light. Chemical mechanics + quantum mechanics = sub elds of mechanics.