CHEM 107 Lecture Notes - Lecture 5: International System Of Units, Ideal Gas, Torr
31 views2 pages
8 Apr 2016
School
Department
Course
Professor
Document Summary
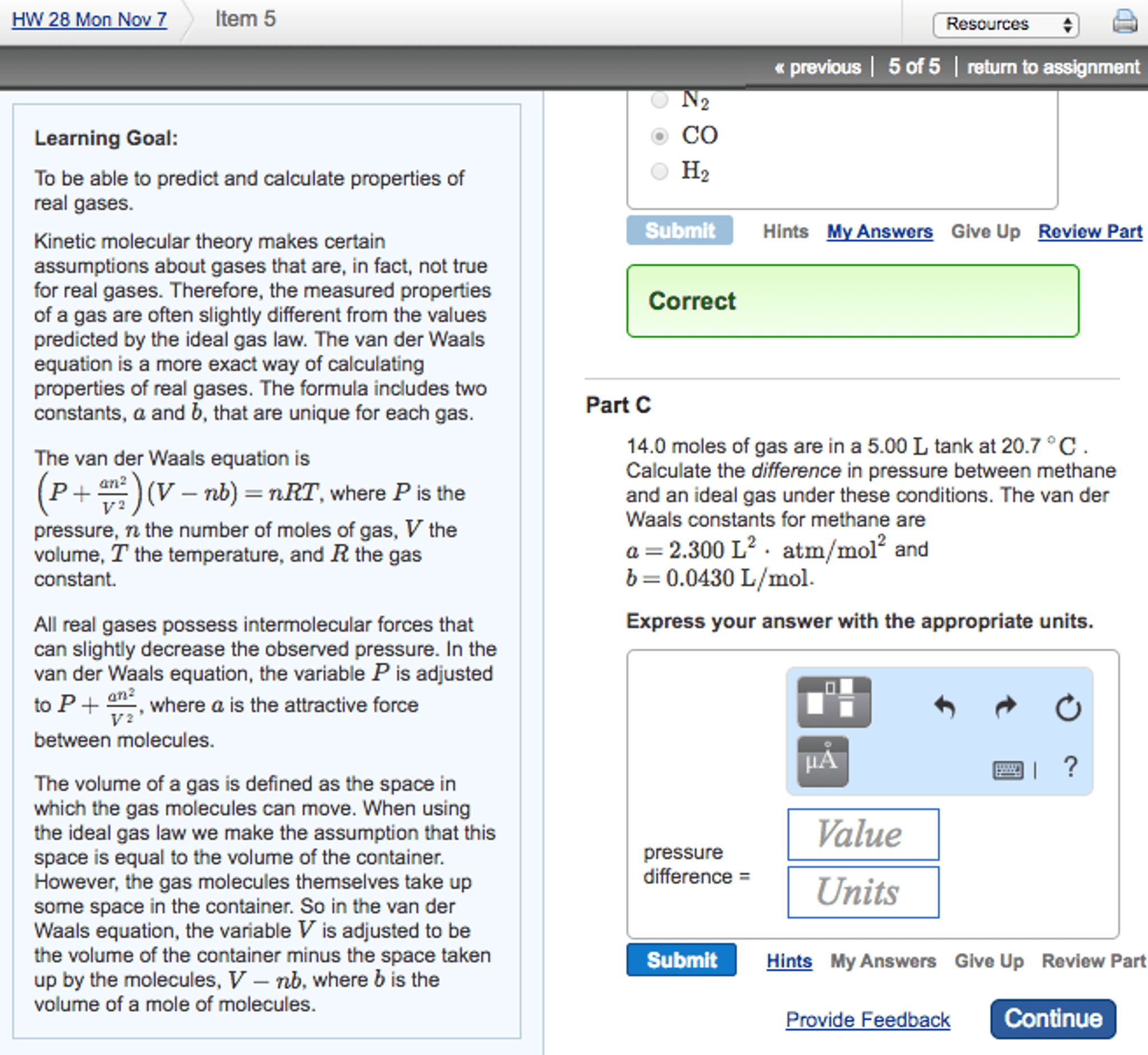
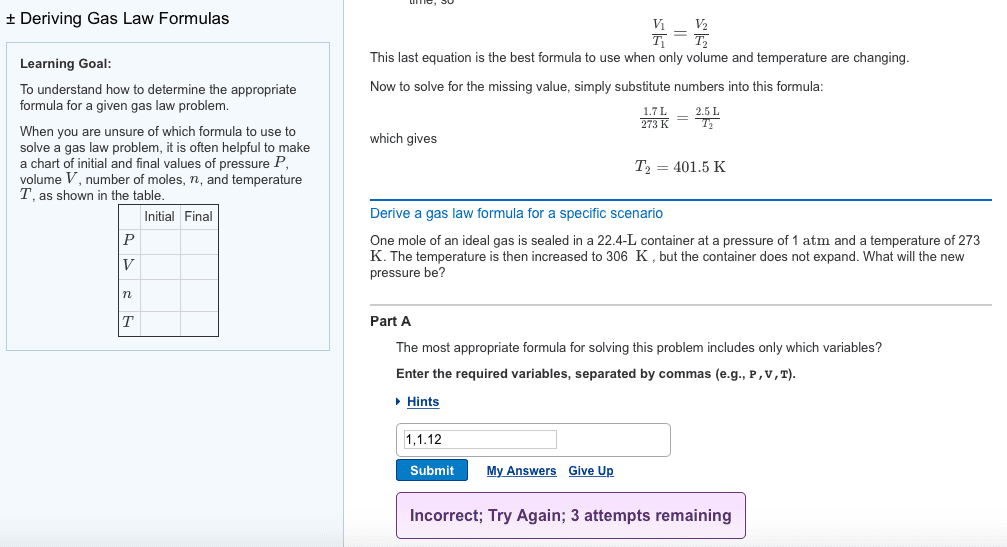
Properies of gases: a gas has no deinite volume or deinite shape but ills the container in which it is in, gases have very low densiies, gas densiies vary with condiions, gases change volume dramaically with changing temperature. V is volume n is moles of gas. T is temperature: originally based on empirical observaions, can be derived mathemaically from the kineic theory of gases. The gas constant r: a proporionality constant. Found out by experiment: in si units. 62. 36 l torr mol-1 k-1: use any convenient units for p and v, must use kelvin and not celsius. Pressure: pressure arises from a huge number of collisions of molecules with walls that contain it, pressure = force/area, unit= pascal.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232