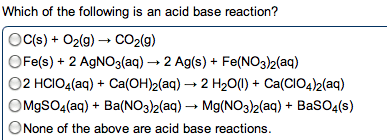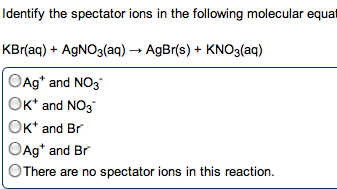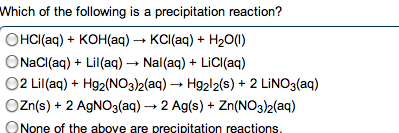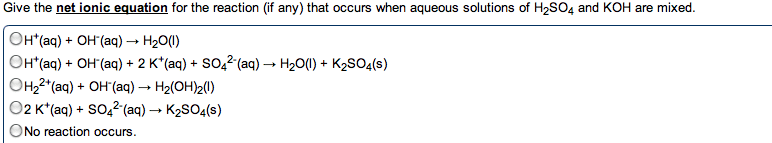CHE 106 Lecture Notes - Lecture 10: Molar Concentration, Acid Strength, Oxidizing Agent
48 views3 pages
3 Oct 2017
School
Department
Course
Professor

63
CHE 106 Full Course Notes
Verified Note
63 documents
Document Summary
Acids : anything that can donate a proton (h+) when associated in a solution. Bases : anything that generates (oh-) in solution (accept protons) The h+ (aq) ions made by acids quickly associate with water molecules to form. Strong acid : an acid that dissociates completely in solution. Weak base : only partially dissociate in solution. When you write an ionic eq. , ions. Neutralization reactions : a reaction between an acid and a base, resulting in the equations formation of a salt (and sometimes water) Strong acid / strong base reaction (example) : : h + (aq) + cl - (aq) + k + (aq) + oh - (aq) k + (aq) + cl - (aq) + h 2 o(l) Hcn(aq) + k+ (aq) kcn (aq) + h2o(l) Ionic: hcn(aq)+ k + (aq) + oh - (aq) k + (aq) + cn - (aq) + h 2 o(l) Carbonates and bicarbonates don"t produce the expected metathesis products.