01:160:162 Lecture 22: Ksp and Molar Solubility
Document Summary
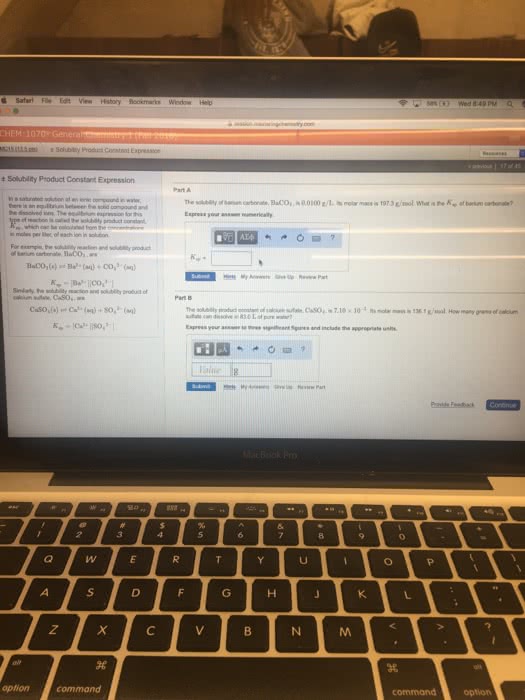
Solubility rules (of ionic compounds in water) allow us to determine ionic compound"s water solubility. Compounds were categorized as either soluble or insoluble. But there are degrees of solubility sparingly soluble, etc. Quantifying solubility of sparingly soluble compound ksp. Sparingly soluble ionic compounds can dissolve in water to some extent. Dissolution of an ionic compound is an equilibrium process. Extent of dissolution can be quantified by an equilibrium constant ex. Solubility of caf2 (s) in water can be represented by: Caf2 (s) ca2+ (aq) + 2 f- (aq) Caf2 (s) + h2o (l) ca2+ (aq) + 2 f- (aq) or. Note** water may or may not be shown in overall reaction. Equilibrium constant for this reaction has a very specific name and equilibrium expression: Remember** pure solids and h2o (l) don"t show up in equilibrium expression. Ksp is a rough measure of compound"s solubility.