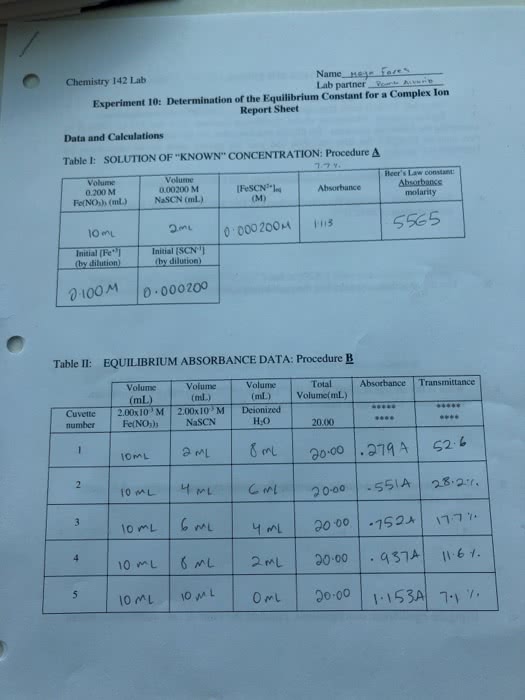
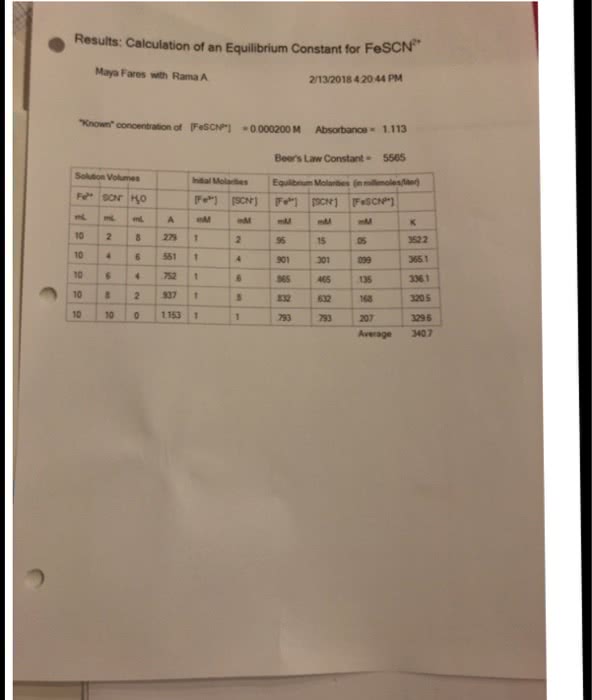
CHE 110L Lecture Notes - Lecture 6: Limiting Reagent, Calcium Chloride, Sodium Hydroxide
Document Summary
The purpose of this experiment was to investigate a series of precipitation reactions involving. Group 1a and 2a ions, write net ionic equations for these reactions, and use one of these reactions to synthesize and characterize a sample of chalk. The procedure for this lab can be found on pages 48-50 in general chemistry che110l. Carbon copies of data tables have been attached to the back of this lab report. 0. 110 g na2co3 x 1 mol na2co3 x 1 mol caco3 x 100. 086 g caco3 = 0. 103 g caco3. 105. 988 g na2co3 1 mol na2co3 1 mol caco3. 0. 148 g cacl2 2h2o x 1 mol cacl2 2h2o x 1 mol caco3 x 100. 086 g caco3 =0. 101 g caco3. 147. 014 g cacl2 2h2o 1 mol cacl2 2h2o 1 mol caco3. Limiting reactant is cacl2 2h2o because out of our two reactant masses it produces the least amount of caco3: actual % yield for chalk.