CHEM 101 Lecture Notes - Chlorate, Lead, Beryllium
47 views1 pages
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
Solubility-Product Constants at 298 K
| Compound | Formula | Ksp |
| Aluminium hydroxide | Al(OH)3 | 4.6Ã10â33 |
| Aluminium phosphate | AlPO4 | 9.84Ã10â21 |
| Antimony sulfide | Sb2S3 | 1.6Ã10â93 |
| Arsenic sulfide | As2S3 | 1Ã10â16 |
| Barium bromate | Ba(BrO3)2 | 2.43Ã10â4 |
| Barium carbonate | BaCO3 | 2.58Ã10â9 |
| Barium chromate | BaCrO4 | 1.17Ã10â10 |
| Barium fluoride | BaF2 | 1.84Ã10â7 |
| Barium hydroxide octahydrate | Ba(OH)2·8H2O | 2.55Ã10â4 |
| Barium iodate | Ba(IO3)2 | 4.01Ã10â9 |
| Barium iodate monohydrate | Ba(IO3)2·H2O | 1.67Ã10â9 |
| Barium molybdate | BaMoO4 | 3.54Ã10â8 |
| Barium nitrate | Ba(NO3)2 | 4.64Ã10â3 |
| Barium phosphate | Ba3(PO4)2 | 3.40Ã10â23 |
| Barium selenate | BaSeO4 | 3.40Ã10â8 |
| Barium sulfate | BaSO4 | 1.08Ã10â10 |
| Barium sulfite | BaSO3 | 5.0Ã10â10 |
| Beryllium hydroxide | Be(OH)2 | 6.92Ã10â22 |
| Bismuth arsenate | BiAsO4 | 4.43Ã10â10 |
| Bismuth iodide | BiI | 7.71Ã10â19 |
| Cadmium arsenate | Cd3(AsO4)2 | 2.2Ã10â33 |
| Cadmium carbonate | CdCO3 | 1.0Ã10â12 |
| Cadmium fluoride | CdF2 | 6.44Ã10â3 |
| Cadmium hydroxide | Cd(OH)2 | 7.2Ã10â15 |
| Cadmium iodate | Cd(IO3)2 | 2.5Ã10â8 |
| Cadmium oxalate trihydrate | CdC2O4·3H2O | 1.42Ã10â8 |
| Cadmium phosphate | Cd3(PO4)2 | 2.53Ã10â33 |
| Cadmium sulfide | CdS | 1Ã10â27 |
| Caesium perchlorate | CsClO4 | 3.95Ã10â3 |
| Caesium periodate | CsIO4 | 5.16Ã10â6 |
| Calcium carbonate (calcite) | CaCO3 | 3.36Ã10â9 |
| Calcium carbonate (aragonite) | CaCO3 | 6.0Ã10â9 |
| Calcium fluoride | CaF2 | 3.45Ã10â11 |
| Calcium hydroxide | Ca(OH)2 | 5.02Ã10â6 |
| Calcium iodate | Ca(IO3)2 | 6.47Ã10â6 |
| Calcium iodate hexahydrate | Ca(IO3)2·6H2O | 7.10Ã10â7 |
| Calcium molybdate | CaMoO | 1.46Ã10â8 |
| Calcium oxalate monohydrate | CaC2O4ÃH2O | 2.32Ã10â9 |
| Calcium phosphate | Ca3(PO4)2 | 2.07Ã10â33 |
| Calcium sulfate | CaSO4 | 4.93Ã10â5 |
| Calcium sulfate dihydrate | CaSO4·2H2O | 3.14Ã10â5 |
| Calcium sulfate hemihydrate | CaSO4·0.5H2O | 3.1Ã10â7 |
| Chromium(III) hydroxide | Cr(OH)3 | 3Ã10â29 |
| Cobalt(II) arsenate | Co3(AsO4)2 | 6.80Ã10â29 |
| Cobalt(II) carbonate | CoCO3 | 1.4Ã10â13 |
| Cobalt(II) hydroxide (blue) | Co(OH)2 | 5.92Ã10â15 |
| Cobalt(III) hydroxide | Co(OH)3 | 1.6Ã10â44 |
| Cobalt(II) iodate dihydrate | Co(IO3)2·2H2O | 1.21Ã10â2 |
| Cobalt(II) phosphate | Co3(PO4)2 | 2.05Ã10â35 |
| Cobalt(II) sulfide | CoS | 4.0Ã10â21 |
| Copper(I) bromide | CuBr | 6.27Ã10â9 |
| Copper(II) carbonate | CuCO3 | 1.4Ã10â10 |
| Copper(I) chloride | CuCl | 1.72Ã10â7 |
| Copper(I) cyanide | CuCN | 3.47Ã10â20 |
| Copper(I) hydroxide | CuOH | 2Ã10â15 |
| Copper(I) iodide | CuI | 1.27Ã10â12 |
| Copper(I) thiocyanate | CuSCN | 1.77Ã10â13 |
| Copper(II) arsenate | Cu3(AsO4)2 | 7.95Ã10â36 |
| Copper(II) hydroxide | Cu(OH)2 | 1.1Ã10â15 |
| Copper(II) iodate monohydrate | Cu(IO3)2·H2O | 6.94Ã10â8 |
| Copper(II) oxalate | CuC2O4 | 4.43Ã10â10 |
| Copper(II) phosphate | Cu3(PO4)2 | 1.40Ã10â37 |
| Copper(II) sulfide | CuS | 6.3Ã10â26 |
| Copper(I) sulfide | Cu2S | 2.5Ã10â48 |
| Europium(III) hydroxide | Eu(OH)3 | 9.38Ã10â27 |
| Gallium(III) hydroxide | Ga(OH)3 | 7.28Ã10â36 |
| Iron(II) carbonate | FeCO3 | 3.13Ã10â11 |
| Iron(II) fluoride | FeF2 | 2.36Ã10â6 |
| Iron(II) hydroxide | Fe(OH)2 | 4.87Ã10â17 |
| Iron(II) sulfide | FeS | 1.6Ã10â19 |
| Iron(III) hydroxide | Fe(OH)3 | 2.79Ã10â39 |
| Iron(III) phosphate dihydrate | FePO4·2H2O | 9.91Ã10â16 |
| Lanthanum iodate | La(IO3)3 | 7.50Ã10â12 |
| Lead(II) bromide | PbBr2 | 6.60Ã10â6 |
| Lead(II) carbonate | PbCO3 | 7.40Ã10â14 |
| Lead(II) chloride | PbCl2 | 1.70Ã10â5 |
| Lead(II) chromate | PbCrO4 | 3Ã10â13 |
| Lead(II) fluoride | PbF2 | 3.3Ã10â8 |
| Lead(II) hydroxide | Pb(OH)2 | 1.43Ã10â20 |
| Lead(II) iodate | Pb(IO3)2 | 3.69Ã10â13 |
| Lead(II) iodide | PbI2 | 9.8Ã10â9 |
| Lead(II) oxalate | PbC2O4 | 8.5Ã10â9 |
| Lead(II) selenate | PbSeO4 | 1.37Ã10â7 |
| Lead(II) sulfate | PbSO4 | 2.53Ã10â8 |
| Lead(II) sulfide | PbS | 8.9Ã10â29 |
| Lithium carbonate | Li2CO3 | 8.15Ã10â4 |
| Lithium fluoride | LiF | 1.84Ã10â3 |
| Lithium phosphate | Li3PO4 | 2.37Ã10â11 |
| Magnesium ammonium phosphate | MgNH4PO4 | 3Ã10â13 |
| Magnesium carbonate | MgCO3 | 6.82Ã10â6 |
| Magnesium carbonate trihydrate | MgCO3·3H2O | 2.38Ã10â6 |
| Magnesium carbonate pentahydrate | MgCO3·5H2O | 3.79Ã10â6 |
| Magnesium fluoride | MgF2 | 5.16Ã10â11 |
| Magnesium hydroxide | Mg(OH)2 | 5.61Ã10â12 |
| Magnesium oxalate dihydrate | MgC2O4·2H2O | 4.83Ã10â6 |
| Magnesium phosphate | Mg3(PO4)2 | 1.04Ã10â24 |
| Manganese(II) carbonate | MnCO3 | 2.24Ã10â11 |
| Manganese(II) iodate | Mn(IO3)2 | 4.37Ã10â7 |
| Manganese(II) hydroxide | Mn(OH)2 | 2Ã10â13 |
| Manganese(II) oxalate dihydrate | MnC2O4·2H2O | 1.70Ã10â7 |
| Manganese(II) sulfide | MnS | 4.6Ã10â14 |
| Mercury(I) bromide | Hg2Br2 | 6.40Ã10â23 |
| Mercury(I) carbonate | Hg2CO3 | 3.6Ã10â17 |
| Mercury(I) chloride | Hg2Cl2 | 1.43Ã10â18 |
| Mercury(I) fluoride | Hg2F2 | 3.10Ã10â6 |
| Mercury(I) iodide | Hg2I2 | 5.2Ã10â29 |
| Mercury(I) oxalate | Hg2C2O4 | 1.75Ã10â13 |
| Mercury(I) sulfate | Hg2SO4 | 6.5Ã10â7 |
| Mercury(I) thiocyanate | Hg2(SCN)2 | 3.2Ã10â20 |
| Mercury(II) bromide | HgBr2 | 6.2Ã10â20 |
| Mercury(II) hydroxide | Hg(OH)2 | 3.6Ã10â26 |
| Mercury(II) iodide | HgI2 | 2.9Ã10â29 |
| Mercury(II) sulfide | HgS | 4Ã10â53 |
| Mercury(I) sulfide | Hg2S | 1.0Ã10â47 |
| Neodymium carbonate | Nd2(CO3)3 | 1.08Ã10â33 |
| Nickel(II) carbonate | NiCO3 | 1.42Ã10â7 |
| Nickel(II) hydroxide | Ni(OH)2 | 5.48Ã10â16 |
| Nickel(II) iodate | Ni(IO3)2 | 4.71Ã10â5 |
| Nickel(II) phosphate | Ni3(PO4)2 | 4.74Ã10â32 |
| Nickel(II) sulfide | NiS | 1.1Ã10â21 |
| Palladium(II) thiocyanate | Pd(SCN)2 | 4.39Ã10â23 |
| Potassium hexachloroplatinate | K2PtCl6 | 7.48Ã10â6 |
| Potassium perchlorate | KClO4 | 1.05Ã10â2 |
| Potassium periodate | KIO4 | 3.71Ã10â4 |
| Praseodymium hydroxide | Pr(OH)3 | 3.39Ã10â24 |
| Radium iodate | Ra(IO3)2 | 1.16Ã10â9 |
| Radium sulfate | RaSO4 | 3.66Ã10â11 |
| Rubidium perchlorate | RbClO4 | 3.00Ã10â3 |
| Scandium fluoride | ScF3 | 5.81Ã10â24 |
| Scandium hydroxide | Sc(OH)3 | 2.22Ã10â31 |
| Silver(I) acetate | AgCH3COO | 1.94Ã10â3 |
| Silver(I) arsenate | Ag3AsO4 | 1.03Ã10â22 |
| Silver(I) bromate | AgBrO3 | 5.38Ã10â5 |
| Silver(I) bromide | AgBr | 5.35Ã10â13 |
| Silver(I) carbonate | Ag2CO3 | 8.46Ã10â12 |
| Silver(I) chloride | AgCl | 1.77Ã10â10 |
| Silver(I) chromate | Ag2CrO4 | 1.12Ã10â12 |
| Silver(I) cobalticyanide | Ag3Co(CN)6 | 3.9Ã10â26 |
| Silver(I) cyanide | AgCN | 5.97Ã10â17 |
| Silver(I) iodate | AgIO3 | 3.17Ã10â8 |
| Silver(I) iodide | AgI | 8.52Ã10â17 |
| Silver(I) oxalate | Ag2C2O4 | 5.40Ã10â12 |
| Silver(I) phosphate | Ag3PO4 | 8.89Ã10â17 |
| Silver(I) sulfate | Ag2SO4 | 1.20Ã10â5 |
| Silver(I) sulfite | Ag2SO3 | 1.50Ã10â14 |
| Silver(I) sulfide | Ag2S | 3.3Ã10â50 |
| Silver(I) thiocyanate | AgSCN | 1.03Ã10â12 |
| Strontium arsenate | Sr3(AsO4)2 | 4.29Ã10â19 |
| Strontium carbonate | SrCO3 | 5.60Ã10â10 |
| Strontium fluoride | SrF2 | 4.33Ã10â9 |
| Strontium iodate | Sr(IO3)2 | 1.14Ã10â7 |
| Strontium iodate monohydrate | Sr(IO3)2·H2O | 3.77Ã10â7 |
| Strontium iodate hexahydrate | Sr(IO3)2·6H2O | 4.55Ã10â7 |
| Strontium oxalate | SrC2O4 | 5Ã10â8 |
| Strontium phosphate | Sr3(PO4)2 | 1Ã10â31 |
| Strontium sulfate | SrSO4 | 3.44Ã10â7 |
| Thallium(I) bromate | TlBrO3 | 1.10Ã10â4 |
| Thallium(I) bromide | TlBr | 3.71Ã10â6 |
| Thallium(I) chloride | TlCl | 1.86Ã10â4 |
| Thallium(I) chromate | Tl2CrO4 | 8.67Ã10â13 |
| Thallium(I) hydroxide | Tl(OH)3 | 1.68Ã10â44 |
| Thallium(I) iodate | TlIO3 | 3.12Ã10â6 |
| Thallium(I) iodide | TlI | 5.54Ã10â8 |
| Thallium(I) thiocyanate | TlSCN | 1.57Ã10â4 |
| Thallium(I) sulfide | Tl2S | 6Ã10â22 |
| Tin(II) hydroxide | Sn(OH)2 | 5.45Ã10â27 |
| Tin(II) sulfide | SnS | 3.2Ã10â28 |
| Tin(IV) sulfide | SnS2 | 1Ã10â70 |
| Yttrium carbonate | Y2(CO3)3 | 1.03Ã10â31 |
| Yttrium fluoride | YF3 | 8.62Ã10â21 |
| Yttrium hydroxide | Y(OH)3 | 1.00Ã10â22 |
| Yttrium iodate | Y(IO3)3 | 1.12Ã10â10 |
| Zinc arsenate | Zn3(AsO4)2 | 2.8Ã10â28 |
| Zinc carbonate | ZnCO3 | 1.46Ã10â10 |
| Zinc carbonate monohydrate | ZnCO3·H2O | 5.42Ã10â11 |
| Zinc fluoride | ZnF | 3.04Ã10â2 |
| Zinc hydroxide | Zn(OH)2 | 3Ã10â17 |
| Zinc iodate dihydrate | Zn(IO3)2·2H2O | 4.1Ã10â6 |
| Zinc oxalate dihydrate | ZnC2O4·2H2O | 1.38Ã10â9 |
| Zinc selenide | ZnSe | 3.6Ã10â26 |
| Zinc selenite monohydrate | ZnSe·H2O | 1.59Ã10â7 |
| Zinc sulfide (alpha) | ZnS | 2Ã10â25 |
| Zinc sulfide (beta) | ZnS | 3Ã10â23 |
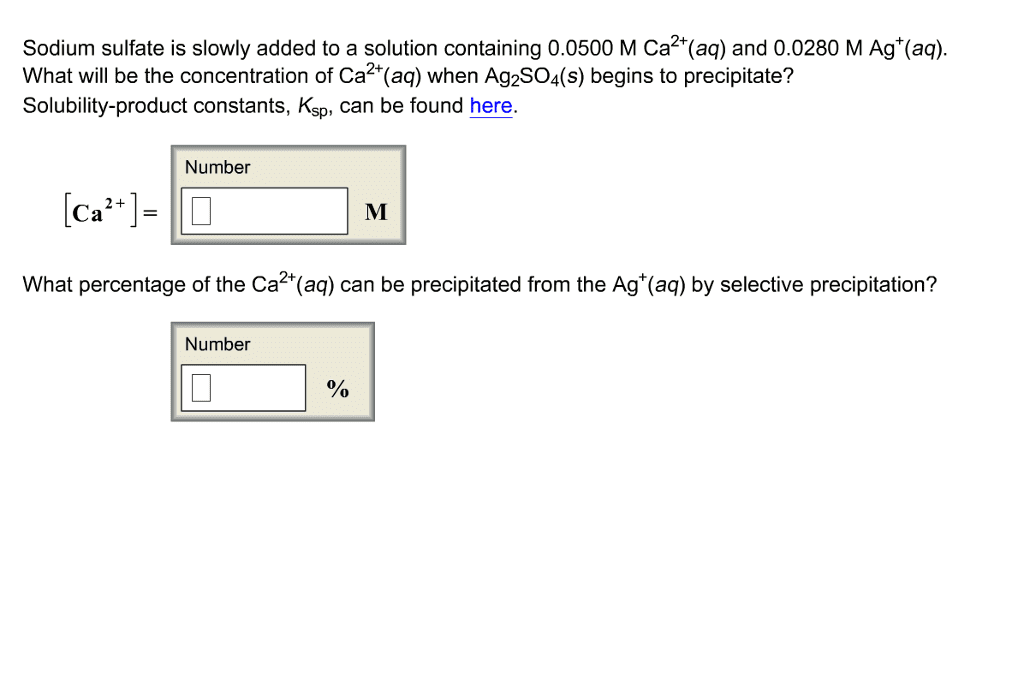
Sodium sulfate is slowly added to a solution containing 0.0500 M Ca^2+(aq) and 0.0280 M Ag^+ (aq). What will be the concentration of Ca^2+(aq) when Ag_2 SO_4 (s) begins to precipitate? Solubility-product constants, K_sp, can be found here. [Ca^2+] = What percentage of the Ca^2+ (aq) can be precipitated from the Ag^+ (aq) by selective precipitation?
