CHEM 1210 Lecture Notes - Lecture 2: Decimal Mark, Significant Figures, Scientific Notation

37
CHEM 1210 Full Course Notes
Verified Note
37 documents
Document Summary
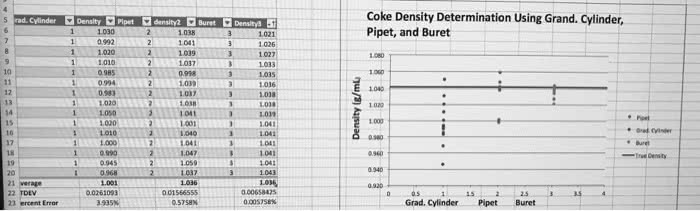
By counting or by definition: 2. 54 cm = 1 in. Precision: how closely individual measurements agree with one another. A(cid:272)(cid:272)ura(cid:272)y: ho(cid:449) (cid:272)losely i(cid:374)di(cid:448)idual (cid:373)easure(cid:373)e(cid:374)ts agree (cid:449)ith the (cid:862)true(cid:863) (cid:448)alue. Significant figures: are the total number of digits in the measurement, the number of digits k(cid:374)o(cid:449)(cid:374) (cid:449)ith (cid:272)ertai(cid:374)ty, plus o(cid:374)e last digit that is the (cid:373)easurer"s (cid:271)est esti(cid:373)ate. The exactness of a measurement is reflected in its sig figs. Zeroes between 2 sig figs are themselves significant. Zeroes at the end of a number to the right of a decimal point are significant. Zeroes at the beginning of a number are never significant. When a number ends in a zero to the left of a decimal point, the zeros are not significant. To clarify ambiguity in rule 5, use scientific notation. Idea: the least accurate value we measure determines the accuracy of the final answer. Addition/subtraction: the last place in the answer is the last place common to all numbers.