CM-UY 1004 Lecture Notes - Lecture 8: Reaction Rate, Fokker E.Ii, Haber Process
12 views8 pages
Document Summary
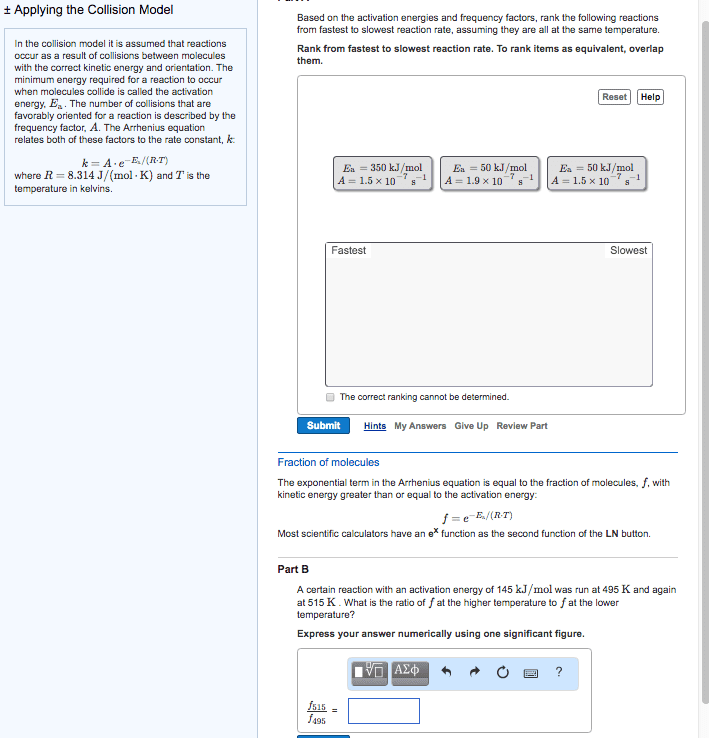
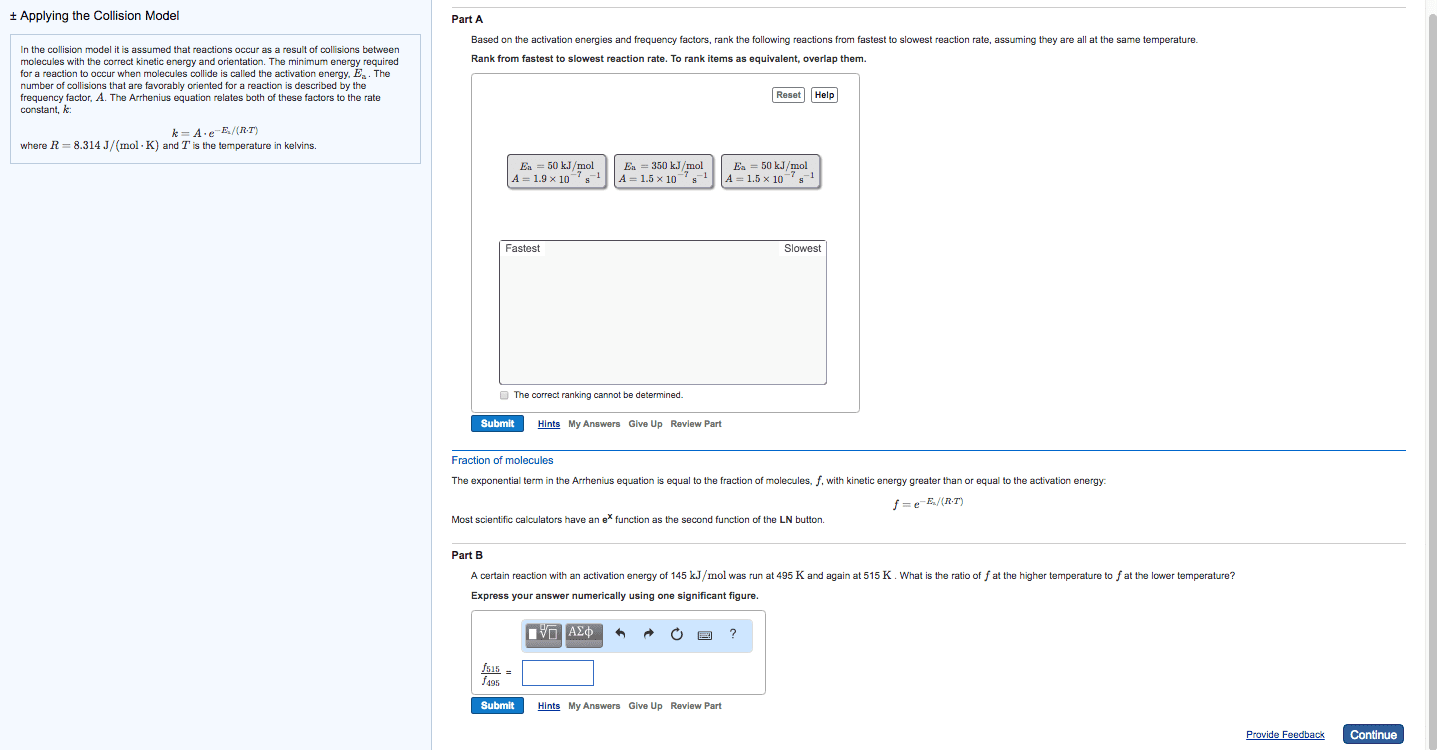
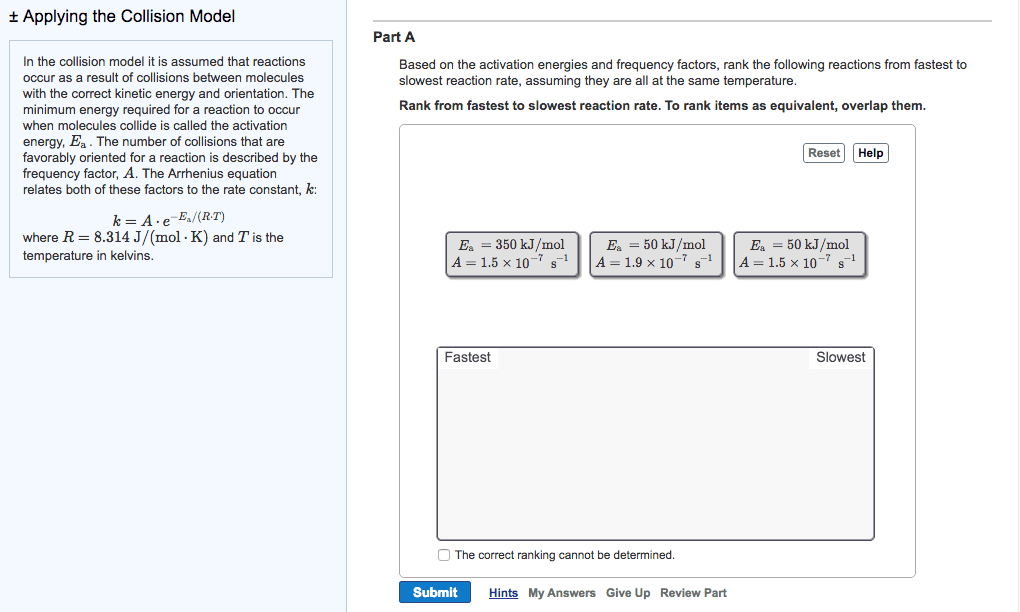
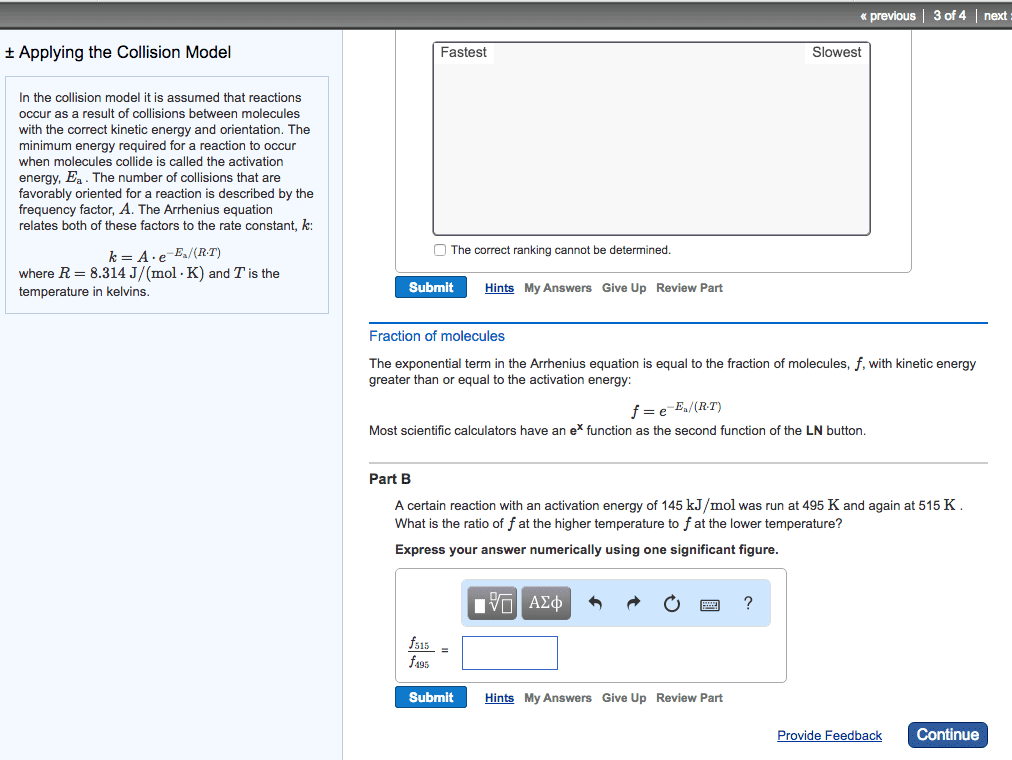
Temperature and rate, rates generally increase as the temperature rises, the collision model b. i. The greater the number of collisions/s the greater the reaction rate b. ii. 1. As reactant concentration increases, the number of collisions increases, leaning to an increase in reaction rate b. iii. Svante arrhenius suggests that molecules must possess a certain minimum amount of energy to react d. ii. This energy comes from the kinetic energies of the colliding molecules d. iii. d. iv. At higher temperature a greater fraction of the molecules have kinetic energy greater than ea, which leads to a greater rate of reaction d. v. d. vi. The fraction of molecules that have an energy equal to or greater than the activation energy is given by: d. vi. 1. Order in which the bonds are broken and formed, and the changes in relative positive of the atoms in the course of the reaction: elementary reactions b. i. Elementary reactions are reactions that occur in a single event or step b. ii.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232