CM-UY 1004 Lecture Notes - Lecture 6: Bromine, Sodium Hydroxide, Sodium Chloride
47 views4 pages
20 Sep 2016
School
Department
Course
Professor
Document Summary
Synthesis (direct combination): element/compound + element/compound one compound. Decomposition (analysis): one compound element/compound + element/compound. Single replacement: element + compound compound + element. Double replacement (exchange of ions): compound + compound compound + compound. Combustion: hydrocarbon + oxygen carbon dioxide + water. Water is always gaseous during a combustion reaction. Exception: 2 ch3oh + o2 4 h2 + 2 co2. Alkali metal + water hydrogen gas + base 2 na + 2 h2o h2 + 2 naoh. Cation: positive charged ion (gets smaller, are metals, and loses electrons) Anion: negative charged ion (gets larger, are nonmetals and gains electrons) Democritus: world is made of empty space and tiny particles atomos . Aristotle: matter is continuous (not made of small particles) false. Boyle: worked with gases (v vs. p) and was the father of modern chemistry. Lavoisier: the law of conservation of mass (matter can"t be created or destroyed, it can only be rearranged)
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
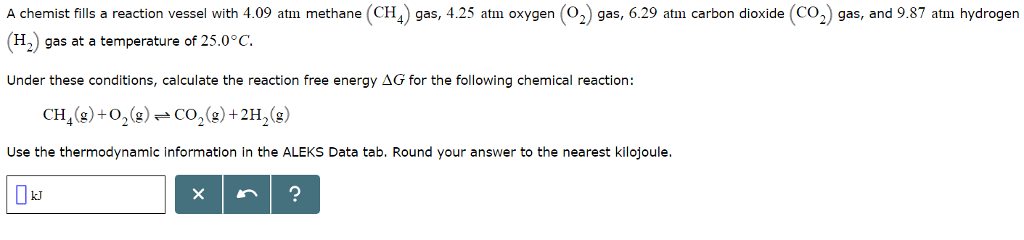
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
A chemist fills a reaction vessel with 4.09 atm methane (CH gas, 4.25 atm oxygen (2) gas, 6.29 atm carbon dioxide (CO2 gas, and 9.87 atm hydrogen (H2) gas at a temperature of 25.0° C. Under these conditions, calculate the reaction free energy ÎG for the following chemical reaction: CH4(g) +02(g)--CO2(g)+2H2(g) Use the thermodynamic information in the ALEKS Data tab. Round your answer to the nearest kilojoule
