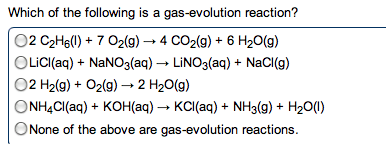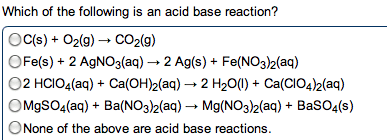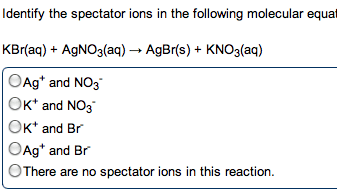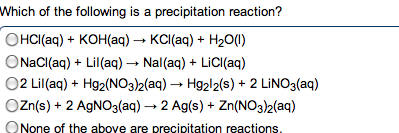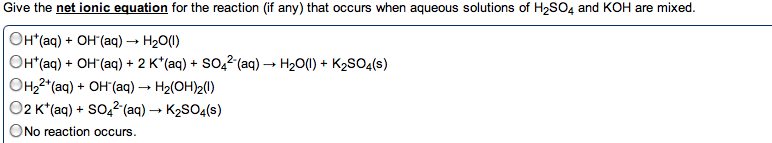CHEM 102 Lecture Notes - Lecture 17: Litmus, Amphoterism, Acid Strength
48 views4 pages
Document Summary
In water, h+ always associates w/ h2o to make h3o (hydronium ion) Acids and bases combine to form h2o, neutralizing the solution. Substance that makes h+ ions in aq solution. Substance that makes oh- ions in aq solution. Concentration of h3o = concentration of strong acid. Ha (aq) h+ (aq) + a- (aq) Produce oh- by accepting a proton from water and ionizing water. Water is amphoteric and can act as an acid or base. Two substances related to eachother by the transfer of a proton. Any base to which a proton has been added. Originally a base but becomes an acid. Any acid to which a proton has been removed. Originally an acid but becomes a base. 3+ 2 4+ . Polarity and strength of bond affect how easily the h is donated. H must be the positive pole to be acidic. Acidity increases right (w/ electronegativity) as bonds become more polar.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232