CHEM 101 Lecture Notes - Lecture 4: Stoichiometry, Endothermic Process, Exothermic Reaction
Document Summary
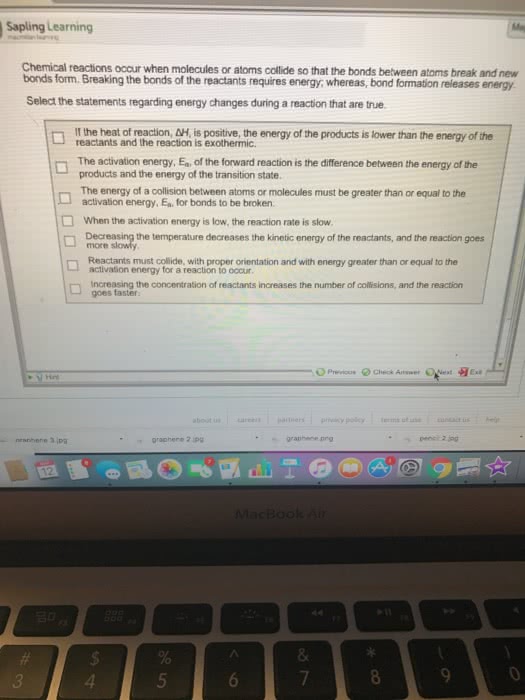
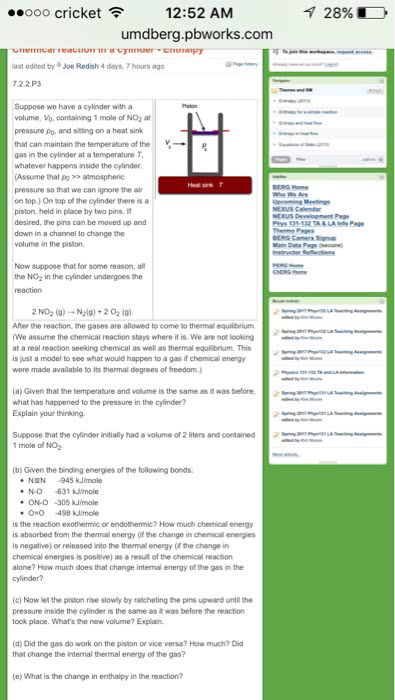
Enthalpy: enthalpy is associated with the stoichiometry of the reaction. Mass, number of moles, number of particles: enthalpy is a state function in terms of the state of the system, enthalpy is the heat (thermal energy change at constant pressure) Bond enthalpy (energy: energy must be supplied to break bonds, in order to overcome stabilization, energy must be put in, higher -> lower energy: bonds made. And triple bonds when calculating enthalpy: h=hreactants-hproducts, weak bonds have less energy while strong bonds have more energy. Be careful about this when calculating h. Calorimetry: the measurement of heat (enthalpy) of chemical and psychical changes, basic principle of calorimetry: energy conservation, put reaction in a bigger system (generally water) then measure the temperature change of that bigger system. Reaction is exothermic, water increases in temperature. Reaction is endothermic, water decreases in temperature. Must keep everything isolated from the rest.