CHEM 101 Lecture Notes - Lecture 1: Rutherford Model, Electromagnetic Radiation, Kinetic Energy
Document Summary
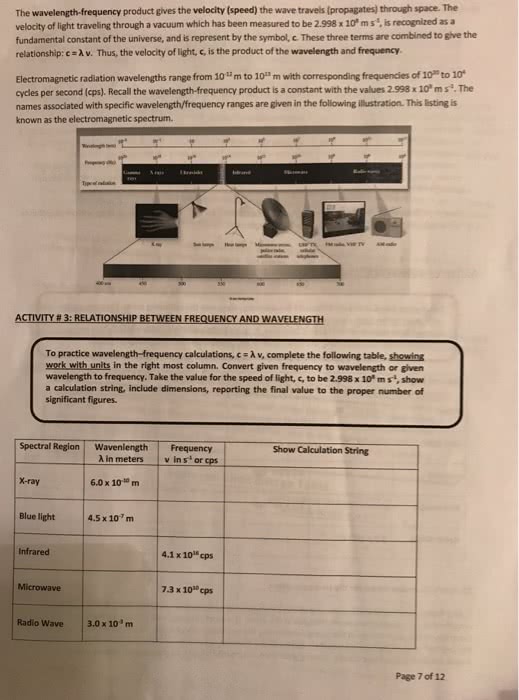
Kinetic energy: has some characteristics of a wave (energy), and some characteristics of a particle (matter). Electromagnetic radiation has two oscillating components, an electric field wave and a magnetic field wave, perpendicular to one another. Amplitude: height of the wave (intensity of the energy) Frequency: number of wavelengths that pass a fixed position in a given number of time (energy of the wave, measured in cycles per second = hertz (hz)) Wavelength: distance between repeating units in the wave (peak to peak, in meters) Wave length and frequency are related through the speed of the wave. Am radio- broadcasted at low energy (amplitude modulation) Fm radio- shorter wave length, higher energy (frequency modulation) Lower energy electromagnetic waves are more easily scattered, leading to am radio waves being able to travel farther as they bounce off the atmosphere or other objects. Fm radios transfer a shorter difference than am radio frequency.