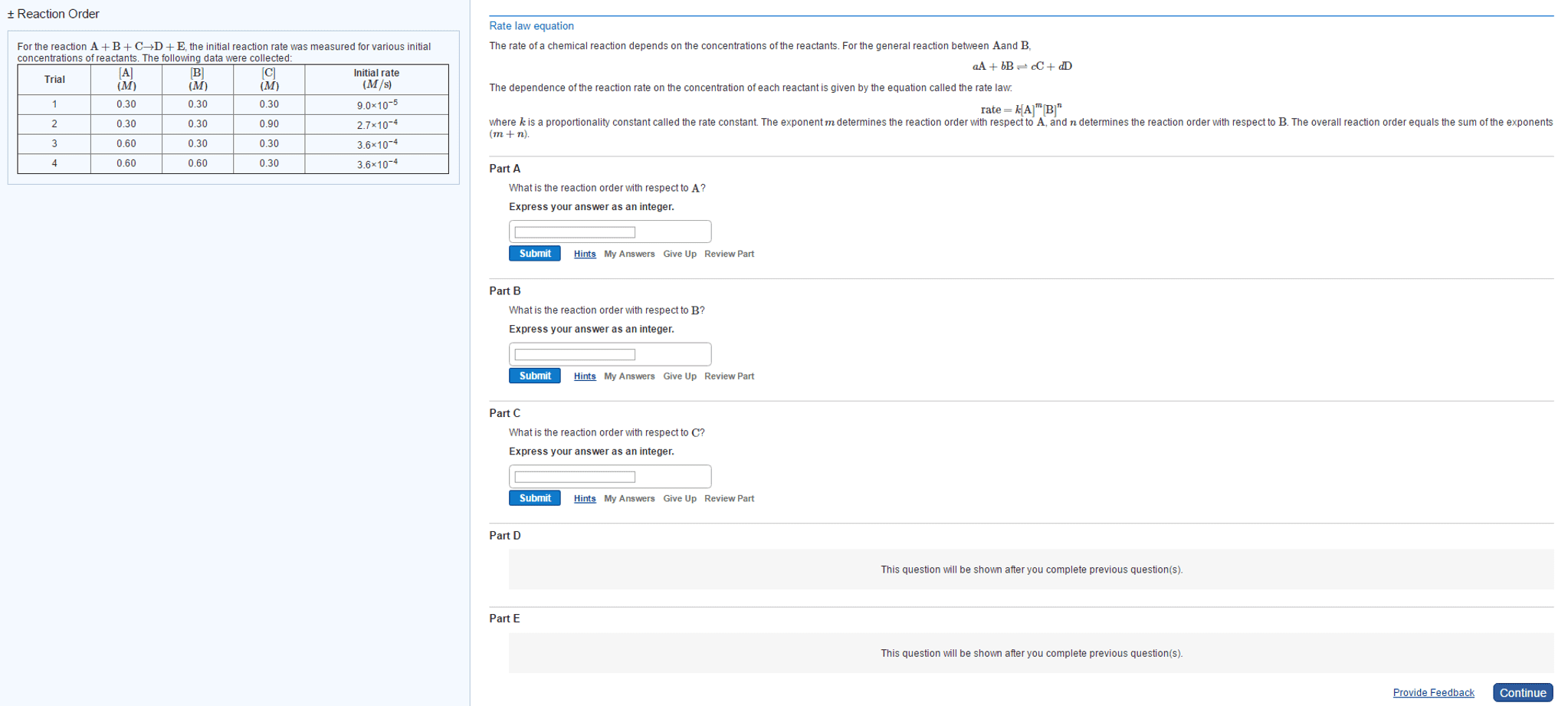
CHEM 1007 Lecture Notes - Lecture 1: Rate Equation, Reaction Rate Constant, Equilibrium Constant
Document Summary
The rate of the reaction is the chance in concentration of reactants (or products) per unit time, units moldm-3s-1. Rate is affected by temperature, concentration, pressure, surface area & catalysts. The order of reaction with respect to a/b is the power of that reactant"s concentration in the rate equation. The overall order of reaction is the sum of orders of all species (i. e add up powers of reactant concentrations) Rate changes with orders of species: 1st order, double conc, double rate, 2nd order, double conc, quadruple rate, 3rd order, double conc, 9 x rate. If a species has zero order, then a change in concentration has no effect on rate. Catalysts may be in the rate equation, and some species in the reaction equation may not be. The units of k depend on the overall order: To find k graphically, plot conc vs time, take tangents and plot rate vs conc.