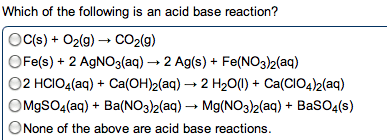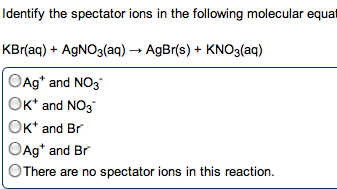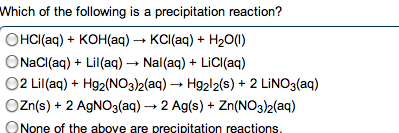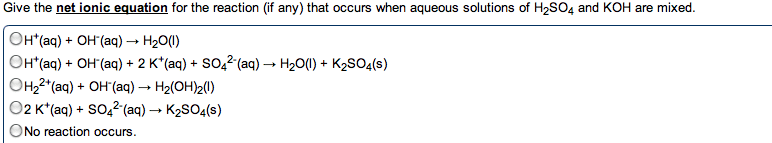CHEM 111 Lecture Notes - Oxidation State, Strong Electrolyte, Spectator Ion
25 views4 pages
Document Summary
Compounds that ionize in solution will conduct electricity. Amount of current related to amount of ionization. Hcl (g) h2o h+ (aq) + cl- (aq) In every acid / base reaction we will consider, the products are water and a salt. Hx (aq) + moh (aq) h2o (l) + mx (aq) Hcl (aq) + na(oh) (aq) nacl (aq) + h2o (l) H+ (aq) + cl- (aq) + na+ (aq) + oh- (aq) na+ (aq) + cl- (aq) + h2o (l) H+ (aq) + oh- (aq) h2o (l) Is there ever a time when the total ionic and net ionic equations are identical: never, sure. If the salt that is formed is insoluble, there are no spectator ions. Redox oxidation-reduction reactions, where there is an exchange of electrons, occurs in both ionic and covalent reactions. Oxidizing agent that which does the oxidizing. Reducing agent that which does the reducing. Assigned to each atom in a formula based on electronegativities.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232