CHEM 130 Lecture Notes - Lecture 5: Proton Nuclear Magnetic Resonance, Carbon-13 Nuclear Magnetic Resonance, Chemical Shift
21 views1 pages
Document Summary
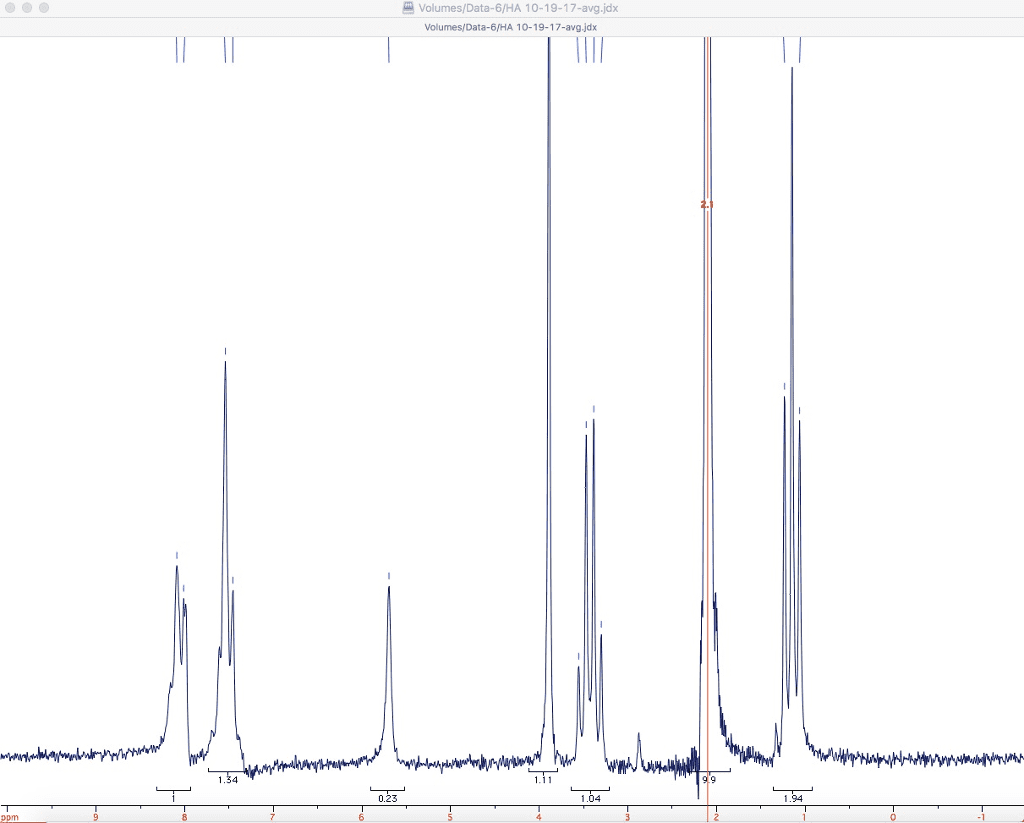
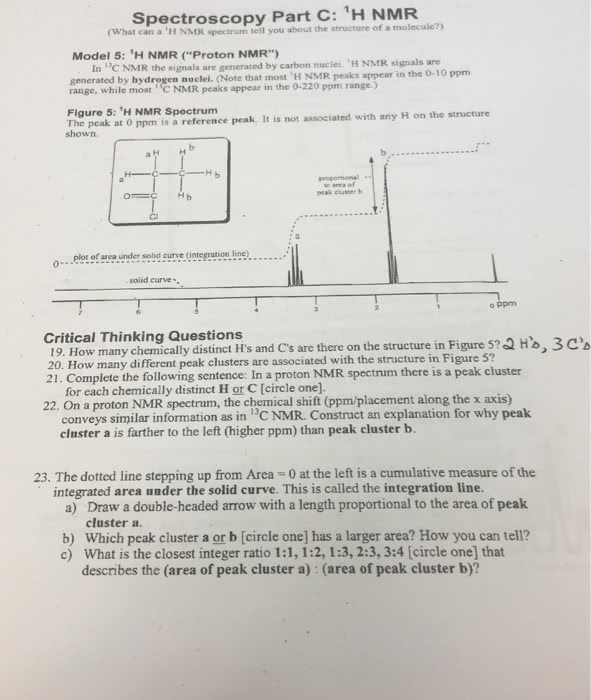
Integration tells us the number of hydrogen atoms in each peak. The chemical shift of the twelve hydrogen atoms of the four identical methyl groups in me4si is defined as zero. The methyl group in the acid is next to the carbonyl group and so slightly deshielded at about d 2. 0 p. p. m. and the acidic proton itself is very deshielded at d 11. 2 p. p. m. The same factor that makes this proton acidic the o h bond is polarized towards oxygen also makes it resonate at low field. So far things are much the same as in carbon nmr. Notice that the ratio of the peak heights in this spectrum was about 3:1 and that that is also the ratio of the number of protons. In fact, it"s not the peak height but the area under the peaks that is exactly proportional to the number of protons.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232