CAS CH 171 Lecture Notes - Lecture 5: Relative Atomic Mass, Chemical Formula, Atomic Number
Document Summary
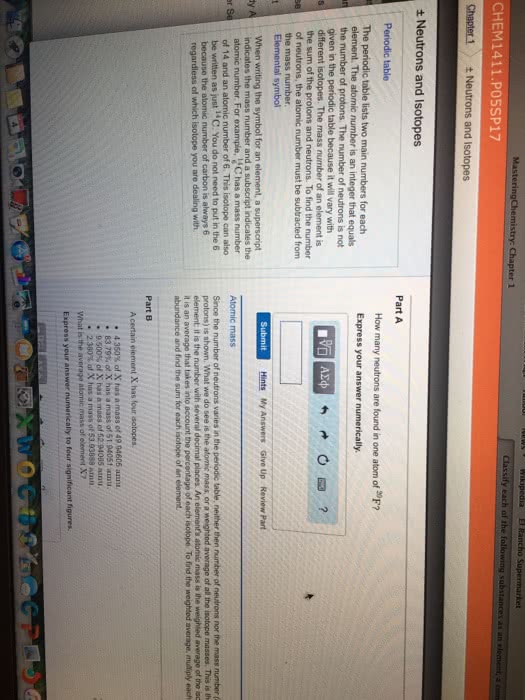
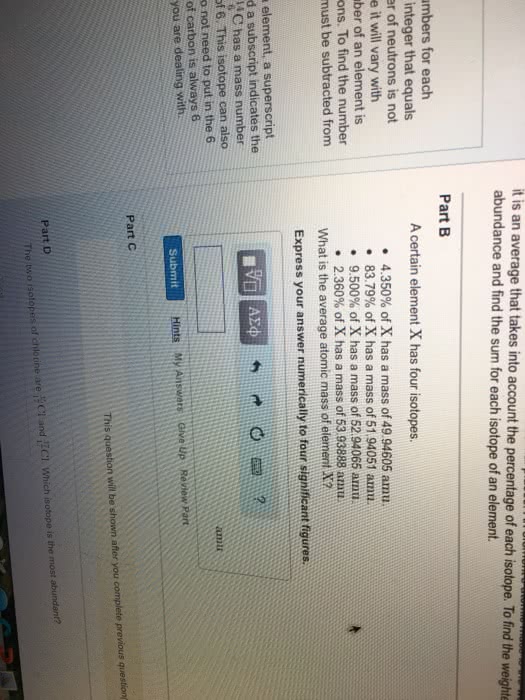
6c 6 is the number of protons= atomic number. 12c 12 is the atomic mass or the number of protons and neutrons isotopes same number of protons and electrons, but different number of neutrons so their atomic masses are different. Relative mass (atomic weight)- really the atomic mass. Atomic mass units (amu)- defined as 1/12 of 12c. Just multiply and add the atomic masses of each atom in the molecular formula. 72. 06 + 12. 0948 + 95. 994 = 180. 1488 180. 15 (round because 5sfs) Weighted average- depends on how often stable isotope is found naturally. The 3 naturally occurring isotopes of hydrogen and their relative abundances: H-1 = 99. 988% h-2 = 0. 01056% and h-3 = . 00144% The atomic weight of each isotope if h-1= 1. 0077u, h-2= 2. 013 u and h-3= 3. 018 u. Percentage shows how often they occur which is why the average weight is closest to the one with the highest percentage. 35. 46 exact conversion so use same number of sfs.