CHEM 1A Lecture Notes - Lecture 15: Hypobromous Acid, Conjugate Acid, Acid Strength
40 views5 pages
Document Summary
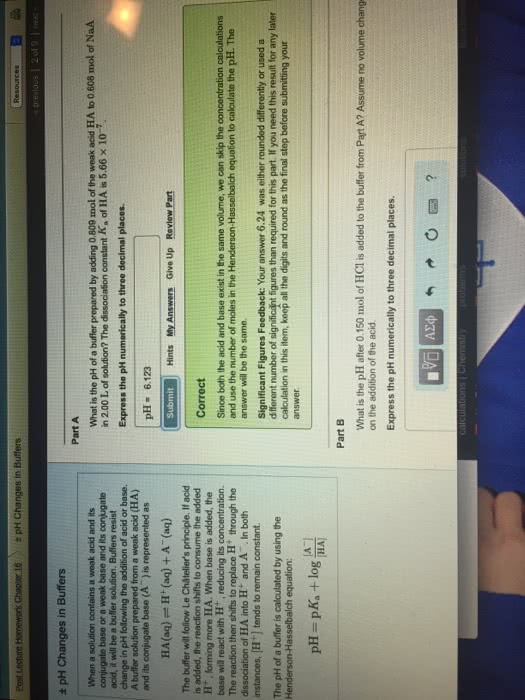
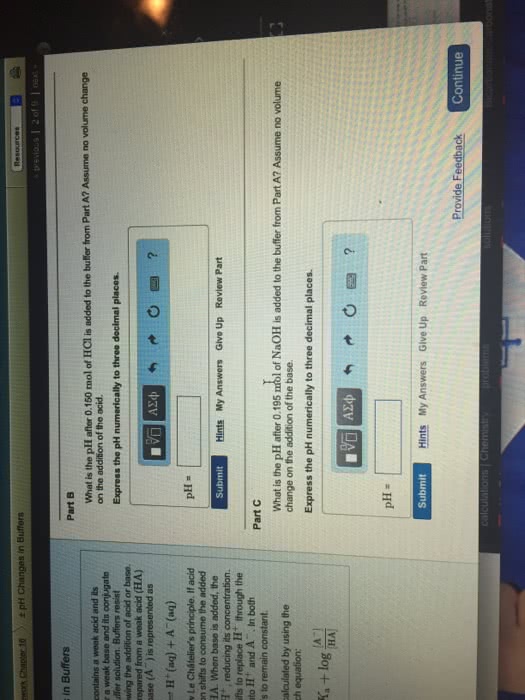
Weak acids and bases, unlike their strong counterparts, do not dissociate fully. The extent of their dissociation is expressed by the magnitude of their dissociation constants, ka and kb, respectively. Expressions of the acid and base dissociation constants can be used to calculate the ph of a solution of a weak acid or base when their initial, analytical concentration is known. One of the dissociation products of a weak acid, ha, is its conjugate base, a-. The basicity of that base is inversely related to the acidity of its conjugate acid, ha. Weak acids generate relatively stronger weak bases and vice versa. The product of the acid and base dissociation constants for every conjugate acid-base pair is constant and is equal to kw. You should be able to: calculate the ph of weak acid and base solutions, calculate the ph of a conjugate base solution knowing its initial concentration and the acid dissociation constant of its conjugate acid.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232