CHEM 1500 Lecture Notes - Lecture 21: Intermediate 1, Nitrogen Dioxide, Nitric Oxide
Document Summary
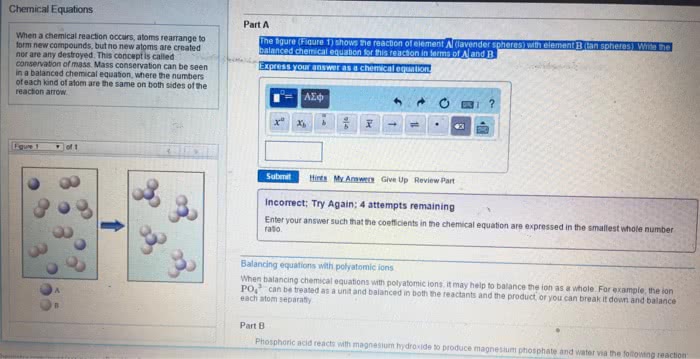
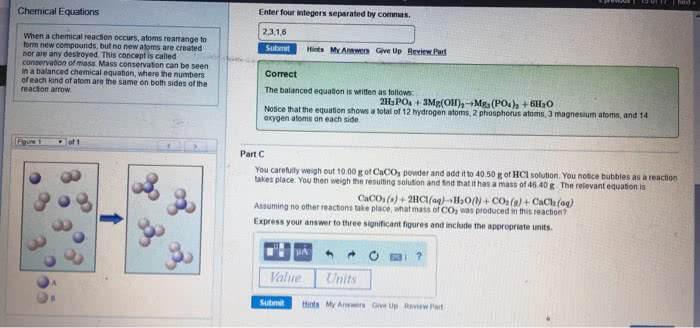
Involves a chemical change (i. e. , change in composition) Is a process in which substance(s) called reactant(s) are converted into new substance(s) called product(s) May show an immediate evidence such as a colour change, precipitate, gas bubbling, etc. May need a confirmation of the composition change by chemical analysis. Note: in many cases, no chemical reaction occurs when compounds are mixed. Contains the reactant(s) on the left and the product(s) on the right. They relate amounts of reactants used and products formed and are used in many different calculations. They also remind us of this important point: Based on the law of the conservation of mass, matter cannot be created or destroyed in a chemical reaction. Before you mix them together, both of the reactants are colourless gases, when you start mixing and heating them, you will see a colour change. A red-brown gas will form and that is a clue that a chemical reaction happened.