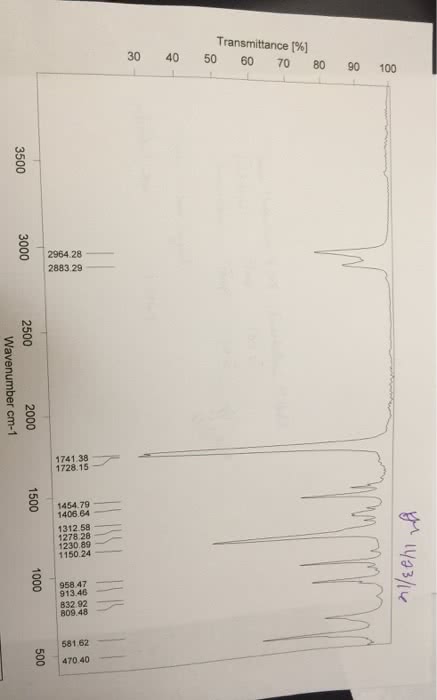
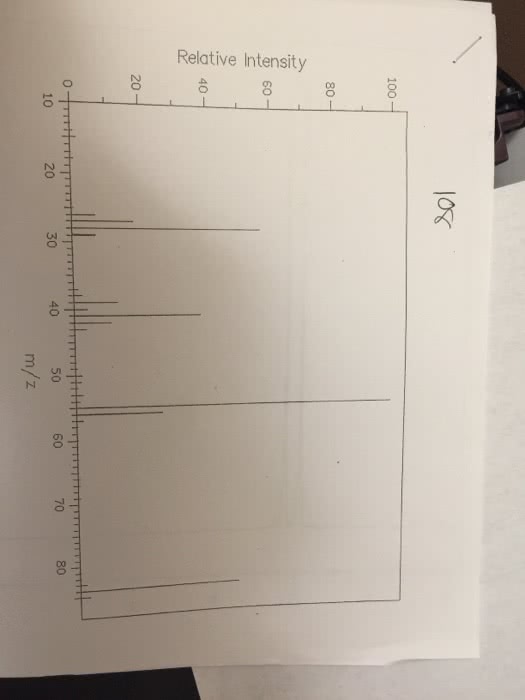
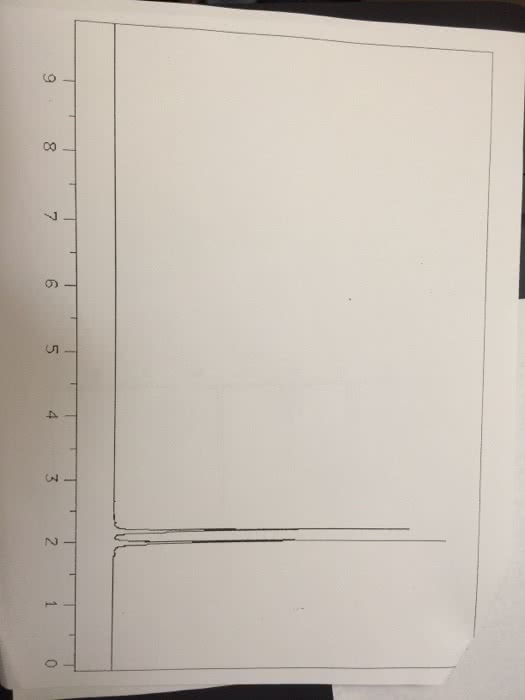
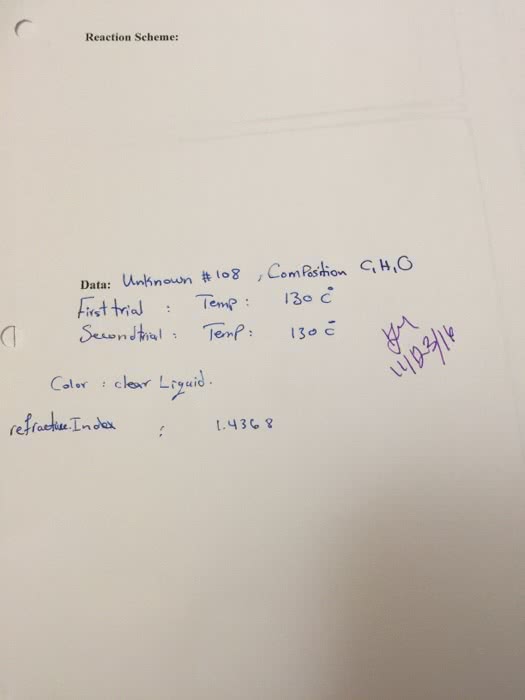Chemistry 1024A/B Lecture Notes - Lecture 1: Atomic Nucleus, Unified Atomic Mass Unit, Atomic Number
Document Summary
The study of matter, its properties and changes at the atomic and molecular level. Matter is anything that has mass and occupies space. Three states: solid: rigid shape and fixed volume, liquid: fixed volume but not rigid in shape, gas: neither fixed volume or rigid shape. Element: a type of matter that cannot be broken down into two or more pure substances. It is a substance in which all atoms have the same atomic number. Compound: a pure substance that contains more than one element. Physical properties: can be observed without changing the chemical identity of the substance. Chemical properties: are observed when a substance takes part in a chemical reaction. Cannot be determined just by viewing or touching the substance. Atom: a neutral species consisting of (atomic nucleus formed from protons and neutrons) Protons- positive charge (+1) and a mass of 1 amu. Electrons- (e-, particles carrying one negative charge (-1), amu of 1/2000.