To separate a mixture of benzoic acid and fluorene, we are going to use an acid-base extraction technique. Most carboxylic acids are insoluble or slightly soluble in water, but they are highly soluble in dilute aqueous sodium hydroxide because the organic acid reacts with the base and produces carboxylate ions, a polar species. In a mixture of a carboxylic acid and an organic compound, the acid may be selectively removed from an organic solvent by extracting with a dilute NaOH solution. After separating the organic and aqueous layers, the carboxylic acid may be recovered from the aqueous solution by adding HCl, which causes precipitation of the carboxylic acid. Draw two structures below.To separate a mixture of benzoic acid and fluorene
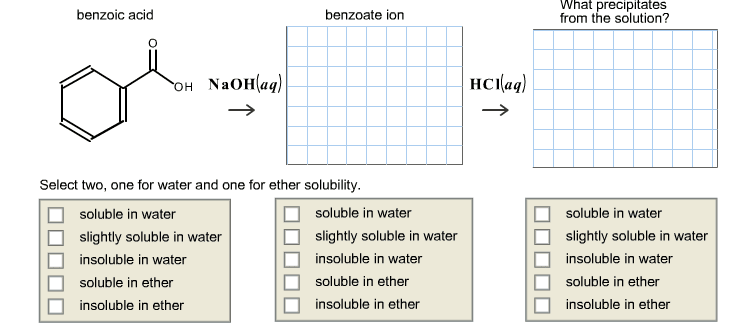
benzoic acid benzoate ion HCI(aq) OH NaOH (aq) Select two, one for water and one for ether solubility soluble in water soluble in water slightly soluble in water slightly soluble in water insoluble in water insoluble in water soluble in ether soluble in ether insoluble in ether insoluble in ether What precipitates from the solution? soluble in water slightly soluble in water insoluble in water soluble in ether insoluble in ether





