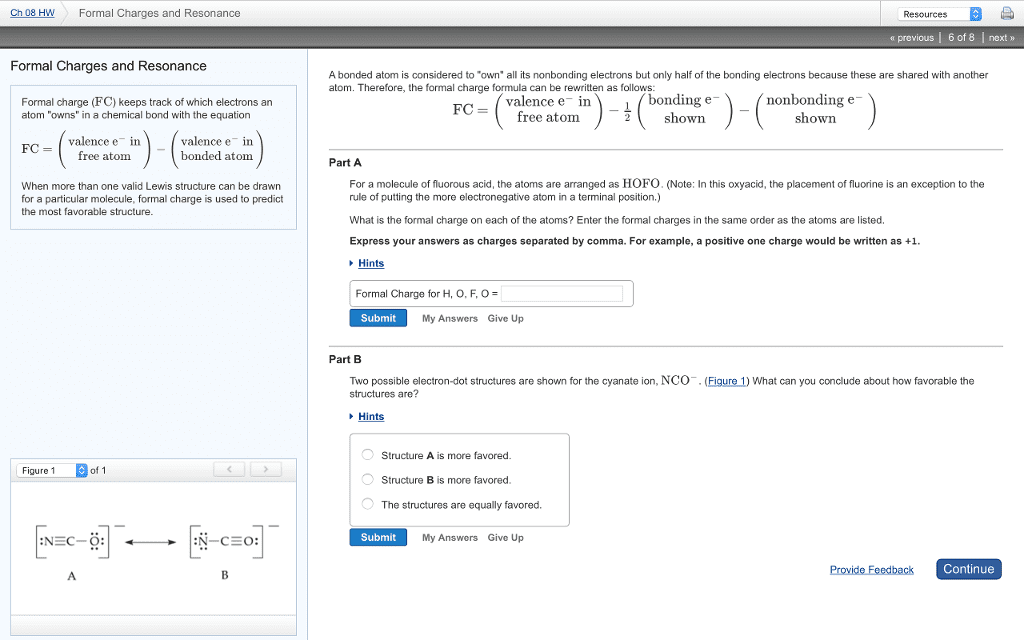
CHM136H1 Lecture Notes - Lecture 7: Formal Charge, Electronegativity

34
CHM136H1 Full Course Notes
Verified Note
34 documents
Document Summary
Drawing resonance structures delta positive charges are given to those that have +1 fc in resonance structures. Since they are electron deficient, they are therefore attacked by electron rich molecules. In decreasing importance: number of bonds. 2. formal charge: charge separation, electronegative atom. 2a: resonance structures with greater charge separation are less important than those with lesser charge separations. Charge separation: fc = largest charge - smallest charge. Electrically neutral = stable e. g. fc of ch3-co-nhr is 1 - (-1) = 2. If comparing the two structures of ch3-co-nhr, if one has charge separation of 0 and the other has 2, with equal number of bonds, the first is favored. The co bond is more double-bond-like, and n-c is more single-bond like. Less important ignore; experimental evidence shows that n is planar, which the first resonance structure does not accurately depict.