CHM135H1 Lecture Notes - Lecture 1: Black-Body Radiation, Electromagnetic Spectrum, Ultraviolet Catastrophe
86 views2 pages

90
CHM135H1 Full Course Notes
Verified Note
90 documents
Document Summary
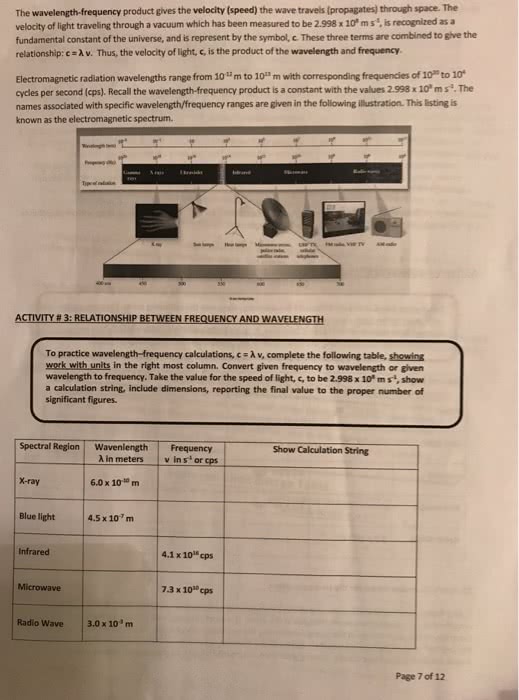
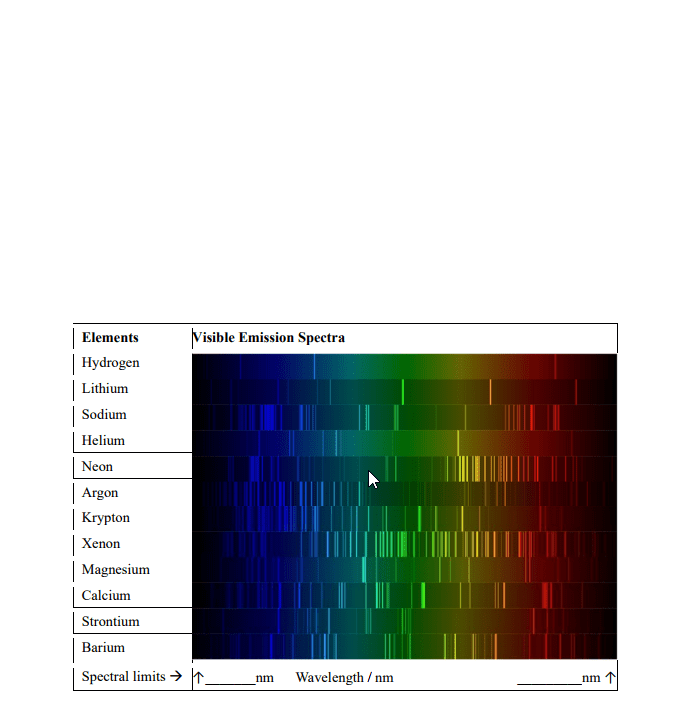
De nition= it is a tabular organization of chemical elements and the most important organizing principle in chemistry. It is ordered by atomic number, electron con guration and chemical properties of elements. The electromagnetic spectrum is a continuous spectrum (made of visible light, ir, uv, radio) The electromagnetic waves (radiation) consist of oscillating electric and magnetic elds perpendicular to each other and direction of propagation: the radiant energy spectrum includes most types of radiations (mostly invisible) b)the visible light spectrum (we can see) Characteristics of electromagnetic energy wavelength( ) = distance light travels in one cycle (m or nm) Frequency(v) = number of wave cycles completed each second (hz) Amplitude(a) = height measured from centre of wave a^2 = intensity. V= c speed of light = 3. 00x10^8m/s. The intensity of brightness depends on amplitude of wave same amplitude= same intensity=same number of photons. Black-body radiation: electromagnetic radiation emitted over wide range of wavelengths when solids are heated.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers