CHM135H1 Lecture 16: 19/09/16 CHM135H1 F LEC0101 Lecture 16 Notes

90
CHM135H1 Full Course Notes
Verified Note
90 documents
Document Summary
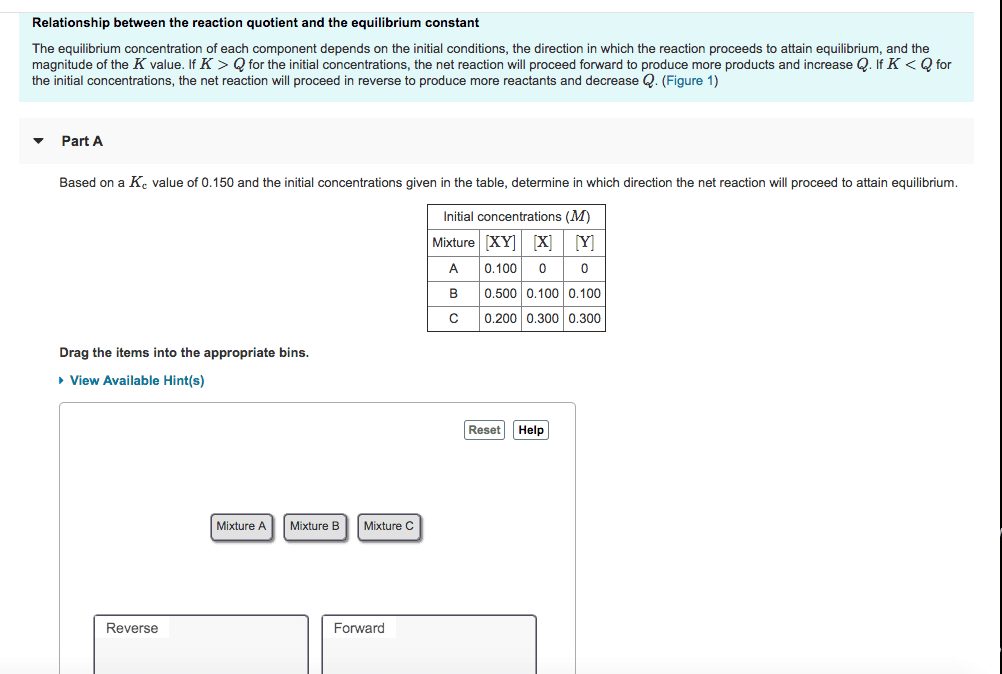
Chm135h1 f - lecture 16 - chapter 14 (1) All reactions will reach a state of equilibrium under suitable conditions. Note that equilibrium does not require that the concentration of each concentration is equal; it is when the concentration stays constant (rate = 0) It does not matter whether the reactant is present initially, or products are present initially - it will still reach the same equilibrium concentration, as long as the conditions are identical. General form: for the reaction aa + bb cc + dd, ]b , where each [ ] is the concentration of the molecule at equilibrium. Kc always has same value, and is independent of initial concentration of reactants and products. Kc is constant for a reaction at a given temperature (and pressure for moles-unbalanced gaseous equations) Concentrations (molarity) of each substance is divided by 1 m, thus concentration units cancel (dimensionless) e. g. k c.