BCH210H1 Lecture 4: Lecture 4

49
BCH210H1 Full Course Notes
Verified Note
49 documents
Document Summary
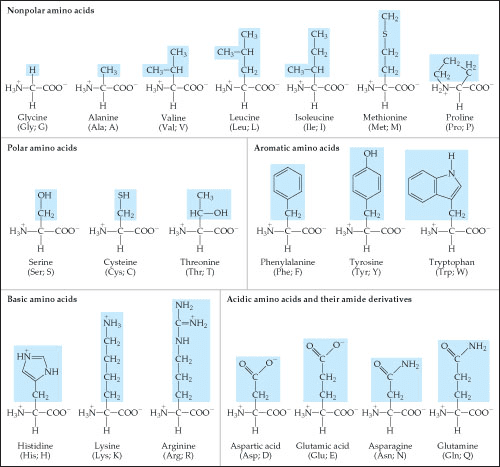
The peptide bond is a linkage that occurs between two amino acids. As a result of the trans linkage, the peptide bond is at or xed on a plane. The dihedral angle about the peptide bond c-n is xed at 180 degrees. Phi is the angle around the n-c alpha bond. Psi is the angle around the calpha-c=o bond. Omega is the angle around the c-n (peptide) bond. Alpha helix phi and psi angle are -60, -50. It can be used to categorize protein and see alpha and beta content. If a protein is needed to be mutated, alanine is a viable candidate as it ts for both alpha and beta helix. In forming a left handed helix, glycine is usually there as it s exible and can t into left handed helix. The type 2 is di erent as the i + one is on the beta.