CHMB41H3 Lecture 2: CHMB41 - Chapter 2
Document Summary
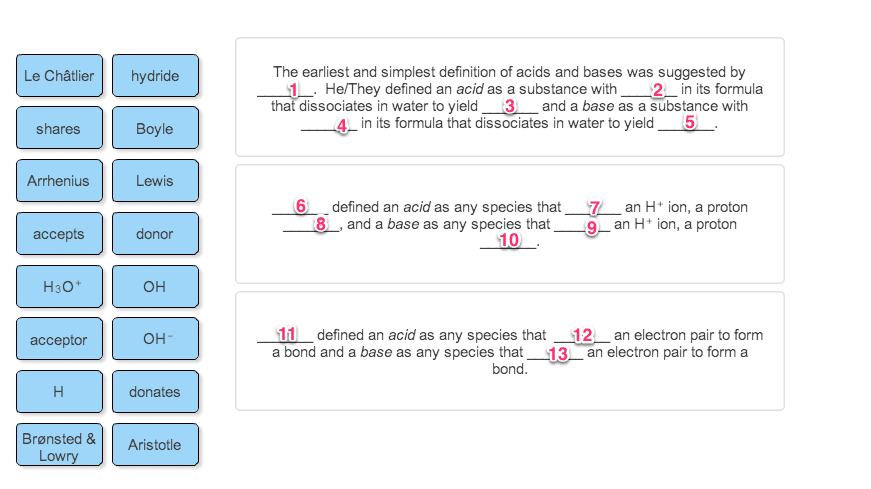
Chapter 2 acids and bases: central to understanding organic chemistry. 2. 1 an introduction to acids and bases. Bronsted-lowry: acid loses a proton, base gains a proton. Any species that has a hydrogen can potentially act as an acid. Any species that has a lone pair can potentially act as a base. The rxn of an acid w/ a base is called an acid-base rxn or a proton transfer rxn. A conjugate base is formed by removing a proton from an acid. A conjugate acid is formed by adding a proton to a base. Acidity is a measure of the tendency of a compound to lose a proton. Basicity is a measure of a compound(cid:495)s affinity for a proton. The stronger the acid, the weaker its conjugate base. The degree to which an acid (ha) dissociates in an aqueous solution is indicated by the equilibrium constant of the rxn, keq.