CHMA11H3 Lecture Notes - Lecture 1: Linear Combination Of Atomic Orbitals, Trigonal Planar Molecular Geometry, Bipyramid
95 views3 pages

36
CHMA11H3 Full Course Notes
Verified Note
36 documents
Document Summary
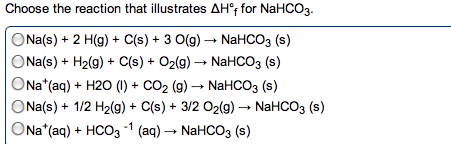
1 mol c3h8: -2044 kj or 5 mol o2: -2044 kj there are 2 mol co2 and 4 mol h2o formed in the reactions. C3h8 (g) + 5 o2 (g) 2 co2 (g) + 4 h2o (g) The magnitude of rh is for the stoichiometric amounts of reactants and products for the reaction as written. A + 2 b c (multiple of reaction) rh 1. 2 a + 4 b 2 c (reverse of reaction) rh 2 = 2 x rh 1. 2 a + 4 b 2 c rh 2 = 2 x rh 1. A + 2 b d h rxn = rh 1 + = rh 4 (sum of reactions) Determining enthalpies of reaction from standard enthalpies of formation. Calculating the standard enthalpy change for a reaction. Standard enthalpy of formation ( rh ) (aka standard heat of formation): for a pure element in its standard state fh = 0. Reactants products rh = rh1 + rh2.