CHMA10H3 Lecture Notes - Lecture 19: Ionic Radius, Electronegativity, Emission Spectrum

36
CHMA10H3 Full Course Notes
Verified Note
36 documents
Document Summary
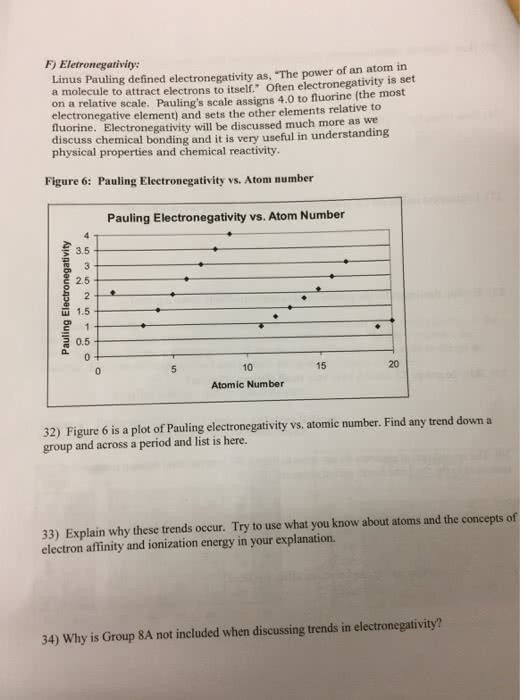
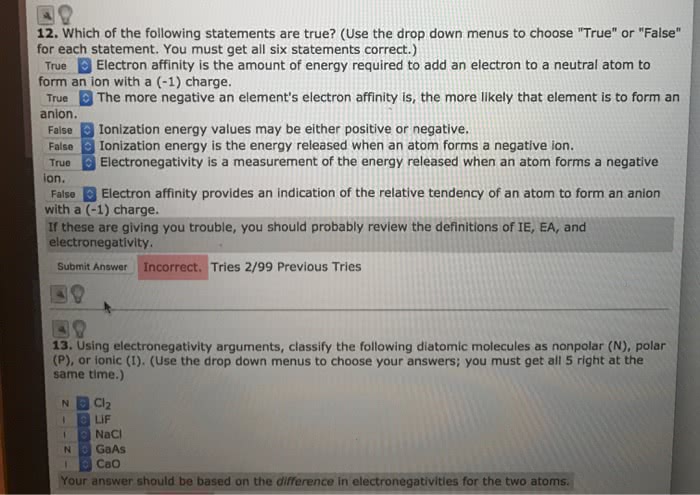
Chma10h3 - introductory to chemistry i: structure and bonding lecture 19: electrons in atoms. If more than one electron can be removes, atoms will have a series of ionization energies: each successive ionization energy (i. e. , second, third, etc. ) is larger than the previous one. Ionic radius: an ionic radius is the estimate based on the distance between the cations and anions in its crystalline compounds, cations are smaller than their parent atoms; anions are larger than their parent atoms. Electron affinity: energy will be released if an electron attaches to an atom to form a negative ion, the electron affinity is the enthalpy change associate with the removal of one electron from the anion. Electrons in an orbit are in a stationary state. This state corresponds to a particular value of energy: while, the electron is in any of its stationary states, it does not radiate energy.