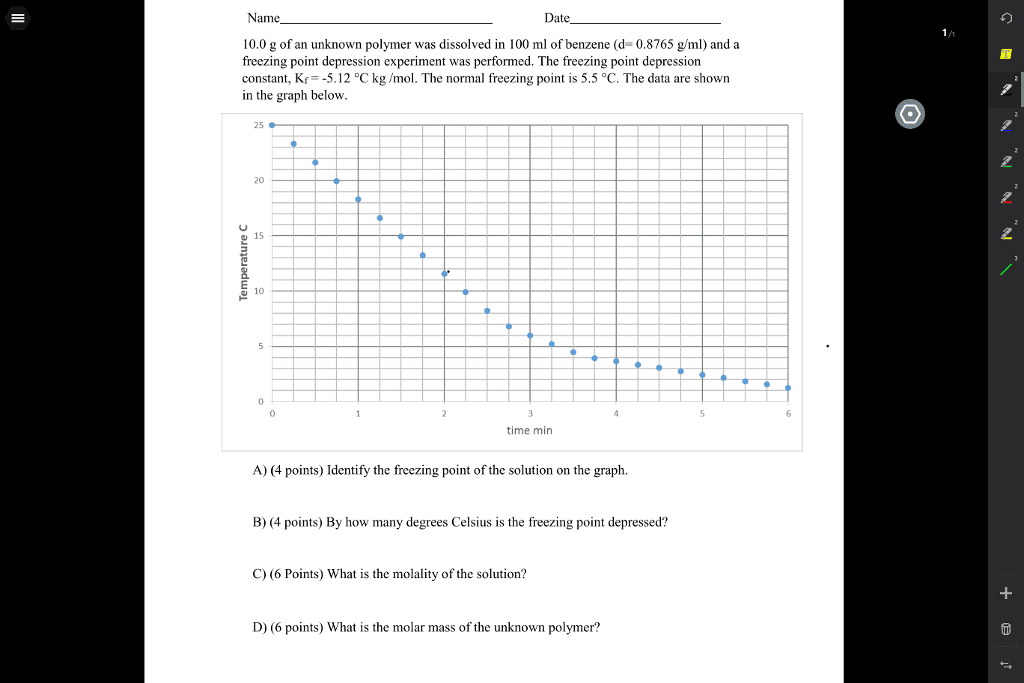
Name Section Advance Study Assignment: Determination of Molar Mass by Depression of the Freezing Point A student determines the molar mass of acetone, CH,COCH, by the method used in this experiment. She found that the equilibrium temperature of a mixture of ice and water was 1.0°C on her thermometer When she added 11.1 g of her sample to the mixture, the temperature, after thorough stirring, fell to -3.00. She then poured off the solution through a screen into a beaker. The mass of the solution was 90.4 g I. a. What was the freezing point depression? b. What was the molality of the acetone? c. How much acetone was in the decanted solution? d. How much water was in the decanted solution? How much acetone would there be in a solution containing 1 kg of water and acetone at the same con- centration as she had in her experiment? e. g acetone r. What did she find to be the molar mass of acetone, assuming she made the calculation properly?